Summary
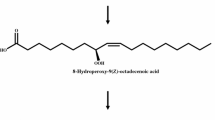
Previously, we reported the discovery of a new compound, 7,10-dihydroxy-8(E)-octadecenoic acid (DOD) which was produced from oleic acid by a new bacterial isolate PR3 [6,7]. The reaction is unique in that it involves a hydroxylation at two positions and a rearrangement of the double bond of the substrate molecule. Now, we have isolated another compound from the reaction mixture determined by GC/MS to be 10-hydroxy-8-octadecenoic acid (HOD). NMR and IR data indicate that the unsaturation is probablycis. The optimum pH and temperature for the production of HOD by strain PR3 were 6.5 and 30°C, about the same as those for DOD. However, the amount of HOD detected remained small throughout an 48-h reaction period during which the amount of DOD increased sharply. At 48 h of reaction, the ratio between HOD∶DOD was 1∶10. HOD may be an intermediate in the biosynthesis of DOD from oleic acid.
Similar content being viewed by others
References
Abbott, B.J. and C.T. Hou. 1973. Oxidation of 1-alkenes to 1,2-epoxyalkanes byPseudomonas oleovorans. Appl. Microbiol. 26: 86–91.
Furuhashi, Keizo 1980. Production of 1,2-epoxides by microorganisms. Japanese Kokai (patent) Sho 55-13.
Furuhashi, K., A. Taoka, S. Ushida, I. Karube and S. Suzuki. 1981. Eur. J. Appl. Microbiol. Biotechnol. 12: 39–45.
Hou, C.T., R.N. Patel and A.I. Laskin. 1982. Microbiological epoxidation of olefin compounds with methylotrophic bacteria conditioned to utilizing methane. U.S. Patent 4, 347, 319.
Hou, C.T., R.N. Patel and A.I. Laskin. 1983. Epoxidation of lower alpha-olefins. U.S. Patent 4, 368, 267.
Hou, C.T., M.O. Bagby, R.D. Plattner and S. Koritala. 1991. A novel compound, 7,10-dihydroxy-8(E)-octadecenoic acid from oleic acid by bioconversion. J. Am. Oil Chem. Soc. 68: 99–101.
Hou, C.T. and M.O. Bagby. 1991. Production of a new compound, 7,10-dihydroxy-8(E)-octadecenoic acid from oleic acid byPseudomonas sp.. PR3. J. Ind. Microbiol. 7: 123–130.
Hou, C.T., L.K. Nakamura, D. Weisleder, R.E. Peterson and M.O. Bagby. 1991. Identification of phenazine-1-carboxylic acid-producing bacterial isolate PR3 which also converts oleic acid to 7,10-dihydroxy-8-octadecenoic acid. Abstract P1, 1991 SIM Annual Meeting, Philadelphia, PA.
Kleiman, R. and G.F. Spencer. 1973. Gas chromatographymass spectrometry of methyl esters of unsaturated oxygenated fatty acids. J. Am. Oil Chem. Soc. 50: 31–38.
Knothe, G.H., M.O. Bagby and C.T. Hou. 1991. 7,10-dihydroxy-8(E)-octadecenoic acid: stereochemical characterization and a novel derivative, 7,10-dihydroxy octadecenoic acid. Abstract. 82nd American Oil Chemist's Society, Annual Meeting. May 12–15, 1991. Chicago, IL.
Koritala, S., L. Hosie, C.T. Hou, C.W. Hesseltine and M.O. Bagby. 1989. Microbial conversion of oleic acid to 10-hydroxystearic acid. Appl. Microbiol. Biotechnol. 32: 299–304.
May, S.W. and B.J. Abbott 1972. Enzymatic epoxidation. I. Alkene epoxidation by the ω-hydroxylation system ofPseudomonas oleovorans. Biochem. Biophys. Res. Commun. 48: 1230–1234.
Niehaus, W.G., Jr. and G.J. Schroepfer, Jr. 1967. J. Am. Chem. Soc. 89:4227.
Wallen, L.L., R.G. Benedict and R.W. Jackson. 1962. The microbiological production of 10-hydroxystearic acid from oleic acid. Arch. Biochem. Biophys. 99:249–253.
Author information
Authors and Affiliations
Additional information
The mention of firm names or trade products does not imply that they are endorsed or recommended by the U.S. Department of Agriculture over other firms or similar products not mentioned.
Rights and permissions
About this article
Cite this article
Hou, C.T., Bagby, M.O. 10-Hydroxy-8(Z)-octadecenoic acid, an intermediate in the bioconversion of oleic acid to 7,10-dihydroxy-8(E)-octadecenoic acid. Journal of Industrial Microbiology 9, 103–107 (1992). https://doi.org/10.1007/BF01569740
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01569740