Abstract
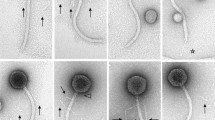
A new virulent bacteriophage, designated RZ1, was isolated from a local pond on the facultative phototrophic bacteriumRhodobacter sphaeroides ZZ101. Electron microscopic studies revealed that, in general morphology, phage RZ1 resembles the λ bacteriophage ofEscherichia coli. The host range of phage RZ1 is limited to some strains ofR. sphaeroides. The phage genome consists of double-stranded DNA of about 44 kb lacking cohesive ends and seems to present terminal redundancy and cyclic permutation. RZ1 phage may carry out a lytic cycle only in recombination-defective mutants ofR. sphaeroides. Nevertheless, a derivative of the RZ1 phage, termed RW1, able to grow in recombination-proficient strains ofR. sphaeroides, has also been obtained. In vitro restriction analysis of both RZ1 and RW1 phages shows the presence of a rearrangement in their DNA. Generalized transduction of Strr and Rifr chromosomal markers has not been detected with either RZ1 or RW1 phages.
Similar content being viewed by others
Literature Cited
Abeliovich A, Kaplan S (1974) Bacteriophages ofRhodopseudomonas spheroides: isolation and characterization of aRhodospeudomonas spheroides bacteriophage. J Virol 13:1392–1399
Adams MH (1959) Bacteriophages. New York: Interscience
Barbé J, Gibert I, Llagostera M, Guerrero R (1987) DNA repair systems in the phototrophic bacteriumRhodobacter capsulatus. J Gen Microbiol 133:961–966
Barbé J, Fernandez de Henestrosa RA, Calero S, Gibert I (1991) Chromogenic method for rapid isolation ofrecA-like mutants of gram-negative bacteria. J Bacteriol 173:404–406
Bradley DE (1967) Ultrastructure of bacteriophages and bacteriocins. Bacteriol Rev 31:230–314
Brooks K, Clark AJ (1967) Behavior of λ bacteriophage in a recombination-deficient strain ofEscherichia coli. J Virol 1:283–288
Calero S, Garí E, Gibert I, Barbé J (1989) Expression of the meta-cleavage pathway operon of the TOL plasmid ofPseudomonas putida in the phototrophic bacteriumRhodobacter sphaeroides. J Biotechnol 12:231–246
Davis RW, Botstein D, Roth JR (1980) Advanced bacterial genetics. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press
Duchrow M, Kohring GW, Giffhorn (1985) Virulence as a consequence of genome instability of a novel temperate bacteriophage, ϕRsG1, ofRhodobacter sphaeroides. Arch Microbiol 142:141–147
Duchrow M, Heitefuss S, Kalkus J, Hoppert M, Giffhorn F (1988) Isolation and characterization of a virulent phage forRhodobacter sphaeroides. Arch Microbiol 149:476–479
Emmerson PT, Howard-Flanders P (1967) Cotransduction withthy of a gene required for genetic recombination inEscherichia coli. J Bacteriol 93:1729–1736
Enquist L, Skalka A (1973) Replication of bacteriophage λ DNA dependent on the function of host and viral genes. I. Interaction ofred, gam andrec. J Mol Biol 75:185–192
Fernandez de Henestrosa AR, Calero S, Barbé J (1991) Expression of therecA gene ofEscherichia coli in several species of gram-negative bacteria. Mol Gen Genet 226:503–506
Fornari CS, Watkins M, Kaplan S (1984) Plasmid distribution and analysis inRhodopseudomonas sphaeroides. Plasmid 11:39–47
Horii ZI, Clark AJ (1973) Genetic analysis of the RecF pathway to genetic recombination inEscherichia coli K-12: isolation and characterization of mutants. J Mol Biol 80:327–333
Llagostera M, Barbé J, Guerrero R (1986) Characterization of SE1, a new general transducing phage ofSalmonella typhimurium. J Gen Microbiol 132:1035–1041
Miller JM (1972) Experiments in molecular genetics. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press
Noland-Jackson E, Miller HI, Adams ML (1978) EcoRI restriction endonuclease cleavage site map of bacteriophage P22 DNA. J Mol Biol 118:347–363
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press
Sistrom WR, Macaluso A, Pledger R (1984) Mutants ofRhodopseudomonas sphaeroides useful in genetic analysis. Arch Microbiol 138:161–165
Smith GR (1983) General recombination. In: Hendrix RW, Roberts JW, Stahl FW, Weisberg RA (eds) Lambda II Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press, pp 175–209
Valentine RC, Shapiro BM, Stadtman ER (1968) Regulation of glutamine synthetase XII. Electron microscopy of the enzyme fromEscherichia coli. Biochemistry 7:2143–2152
Zissler J, Signer ER, Schaefer F (1971) In: Hershey AD (ed) The role of recombination in growth of bacteriophage lambda. I. The gamma gene. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press, pp 455–468
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garí, E., Ollé, M., Gibert, I. et al. Isolation and characterization of a recombination defective-dependent bacteriophage ofRhodobacter sphaeroides . Current Microbiology 24, 151–157 (1992). https://doi.org/10.1007/BF01568980
Issue Date:
DOI: https://doi.org/10.1007/BF01568980