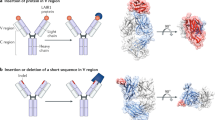
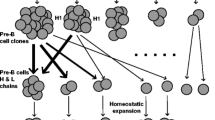
Conclusions
During the last decade, a substantial body of knowledge has been obtained on the generation of somatic diversification of the B cell repertoire, especially with regard to differentiation and selection of B cells in specialized microenvironments such as GCs and GALTs which are similar to each other morphologically and physiologically. Although the mechanisms for somatic diversification remain unclear, with advances in histo- and cytochemical and molecular genetic techniques, some important issues such as the cellular interactions and signals involved in differentiation and selection have been approached and/or identified. Phylogenetic studies will be important to understand relationships between distinct developmental pathways and different lymphoid tissues in various species during evolution.
Biologists will need the reconciliation of three views to gain the real comprehension of immunity: responses of individual cells to intra- and extracellular stimuli, responses of distinct cell populations to each other, and responses of organisms and/or species to their environment in order to survive.
Similar content being viewed by others
References
Eisen HN, Siskind GW: Variation in affinities of antibodies during the immune response. Biochemistry 3:996–1008, 1964
Mitchison NA: The carrier effect in the secondary response to hapten-protein conjugates. II. Cellular cooperation. Eur J Immunol 1:18–27, 1971
Williamson AR, Zitron IM, McMichael AJ: Clones of B lymphocytes, their natural selection and expansion. Fed Proc 35:2195–2201, 1976
Niewenhuis P, Keuning FJ: Germinal centers and the origin of the B cell system. II. Germinal centers in the rabbit spleen and popliteal lymph nodes. Immunology 26:509–519, 1974
MacLennan ICM: Germinal centers. Annu Rev Immunol 12:117–139, 1994
Jacob J, Kelsoe G: In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl II. A common clonal origin for periarteriolar lymphoid sheath-associated foci and germinal centers. J Exp Med 176:679–687, 1992
MacLennan ICM, Lui Y-J, Johnson GD: Maturation and dispersal of B-cell clones during T cell-dependent antibody responses. Immunol Rev 126:143–161, 1992
Grobler P, Buerki H, Cottier H, Hess MW, Stoner RD: Cellular bases for relative ratio resistance of the antibody-forming system at advanced stages of the secondary response to tetanus toxoid in mice. J Immunol 112:2154–2165, 1974
Tsiagbe VK, Linton P-J, Thorbecke GJ: The path of memory B-cell development. Immunol Rev 126:113–141, 1992
Jacob J, Kelsoe G, Rajewsky K, Weiss U: Intraclonal generation of antibody mutants in germinal centers. Nature 354:3898–392, 1991
Berek C, Berger A, Apel M: Mutation of the immune response in germinal centers. Cell 67:1121–1128, 1991
Weiss U, Rajewsky K: The repertoire of somatic antibody mutants accumulating in the memory compartment after primary immunization is restricted through affinity maturation and mirrors that expressed in the secondary response. J Exp Med 172:1681–1698, 1990
Kelsoe G, Zheng B: Sites of B-cell activation in vivo. Curr Opin Immunol 5:418–422, 1993
Zheng B, Xue W, Kelsoe G: Locus-specific somatic hypermutation in germinal center T cells. Nature 370:556–559, 1994
Jacob J, Kassir R, Kelsoe G: In situ studies of the primary response to (4-hydroxy-3-nitrophenyl)acetyl. I. The architecture and dynamics of responding cell populations. J Exp Med 173:1165–1175, 1991
Liu Y-J, Lane PJL, Chan EYT, MacLennan ICM: Sites of specific B cell activation in primary and secondary responses to T cell-dependent and T cell-independent antigens. Eur J Immunol 21:2951–2962, 1991
Klaus GGB, Humphrey JH, Kunkel A, Dongworth DW: The follicular dendritic cell, its role in antigen presentation in the generation of immunological memory. Immunol Rev 53:3–28, 1980
Mandel TE, Phipps RP, Abbot A, Tew JG: The follicular dendritic cells: long term antigen retention during memory. Immunol Rev 53:29–59, 1980
Tew JG, Kosco MH, Burton GF, Szakal AK: Follicular dendritic cells as accessory cells. Immunol Rev 117:185–211, 1990
Schriever F, Nadler LM: The central role of follicular dendritic cells in lymphoid tissues. Adv Immunol 51:243–284, 1992
Fuller KA, Kanagawa O, Nahm M: T cells within germinal centers are specific for the immunizing antigen. J Immunol 151:4505–4512, 1993
Keaney ER, Pape KA, Loh DY, Jenkins MK: Visualization of peptide-specific T cell immunity and peripheral tolerance induction in vivi. Immunity 1:327–339, 1994
McHeyzer-Williams MG, McLean MJ, Lalor PA, Nossal GJV: Antigen-driven B cell diiferentiation in vivo. J Exp Med 178:295–307, 1993
Kroese FGM, Wubennan AS, Seijen HG, Niewenhuis P: Germinal centers develop oligoclonally. Eur J Immunol 17:1069–1072, 1987
MacLennan ICM, Liu Y-J, Oldfield S, Zhang J, Lane PJL: The evolution of B-cell clones. Curr Top Microbiol Immunol 159:37–63, 1990
Zaitoun AM: Cell population kinetics of the germinal centers of lymph nodes of Balb/c mice. J Anat 130:131–137, 1980
Zhang J, MacLennan ICM, Liu Y-J, Lane PJL: Is rapid proliferation in B centroblasts linked to somatic mutation in memory B cell clones? Immunol Lett 18:297–299, 1988
Linton PJ, Decker DJ, Klinman NR: Primary antibody-forming cells and secondary B cells are generated from separate precursor cell subpopulations. Cell 59:1049–1059, 1989
Linton PJ, Lo D, Lai L, Thorbecke GJ, Klinman NR: Among naive precursor cell subpopulations only progenitors of memory B cells originate germinal centers. Eur J Immunol 22:1293–1297, 1992
Liu Y-J, Johnson GD, Gordon J, MacLennan ICM: Germinal centers in T-cell-dependent antibody responses. Immunol Today 13:17–21, 1992
Gray D: Recruitment of virgin B cells into an immune response is restricted to activation outside lymphoid follicles. Immunology 65:73–79, 1988
Vonderheid RH, Hunt SV: Immigration of thoratic duct B lymphocytes into established germinal centers in the rat. Eur J Immunol 20:79–86, 1990
Han SH, Hathcock K, Zheng B, Kepler TB, Hodes R, Kelsoe G: Cellular interaction in germinal centers. Roles of CD40 ligand and B7-2 in established germinal centers. J Immunol 155:556–567, 1995
Jacobson EB, Corporale LH, Thorbecke GJ: Effect of thymus cell injection on germinal center formation in lymphoid tissue of nude mice. Cell Immunol 13:416–430, 1974
Gastkemper NA, Wubbena AS, Gumbrere FJH, DeGraff A, Nieuwenhuis P: Germinal centers and the B-cell system. V. Presence of germinal center-precursor cells among lymphocytes of the thoracic duct in the rat. Cell Tissue Res 219:281–289, 1981
Vonderheide RH, Hunt SV: T lymphocytes in rat germinal centers belong to an ER3+ subpopulation of CD4+ cells. Immunology 69:542–547, 1990
Stedra J, Cerny J: Distinct pathways of B cell differentiation. I. Residual T cells in athymic mice support the development of splenic germinal centers and B cell memory without an induction of antibody. J Immunol 152:1718–1726, 1994
Miller C, Stedra J, Kelsoe G, Cerny J: Facultative role of germinal centers and T cells in the somatic diversification of IgVH genes. J Immunol 181:1319–1331, 1995
Clark EA, Ledbetter JA: How B and T cells talk to each other. Nature. 367:425–428, 1994
Durie FH, Foy TM, Master SR, Lamon JD, Noelle RJ: The role of CD40 in the regulation of humoral and cell-mediated immunity. Immunol Today 15:406–411, 1994
Hathcock KS, Laszlo G, Pucillo C, Linsley P, Hodes RJ: Comparative analysis of B7-1 and B7-2 costimulatory ligands: expression and function. J Exp Med 180:631–640, 1994
Hathcock KS, Laszlo G, Dickler HB, Bradshaw, Linsley P, Hodes RJ: Identification of an alternative CTLA-4 ligand costimulatory for T cell activation. Science 262:905–907, 1993
Freeman GJ, Borriello F, Hodes RJ, Reiser H, Hathcock KS, Laszlo G, McKnight AJ, Kim J, Du L, Lombard DB, Gray GS, Nadler LM, Sharpe AH: Uncovering of functional alternative CTLA-4 counter-receptor in B7-deficient mice. Science 262:907–909, 1993
Renshaw BR, Fanslow WC, Armitage RJ, Campbell KA, Liggitt D, Wright B, Davison BL, Maliszewski CR: Humoral immune responses in CD40 ligand-deficient mice. J Exp Med 180:1889–1900, 1994
Callard RE, Armitage RA, Fanslow WC, Spriggs MK: CD40 ligand and its role in X-linked hyper-IgM syndrome. Immunol Today 14:559–564, 1993
Allen RC, Armitage RJ, Conley ME, Rosenblatt H, Jenkins NA, Copeland NG, Bedell MA, Edelhoff S, Disteche CM, Simoneaux DK, Fanslow WC, Belmont J, Spriggs MK: CD40 ligand gene defects responsible for X-linked hyper-IgM syndrome. Science 259:990–993, 1993
Jabara HH, Fu SM, Geha RS, Vercelli D: CD40 and IgE: synergism between anti-CD40 monoclonal antibody and interleu-kin-4 in the induction of IgE synthesis by highly purified human B cells. J Exp Med 172:1861–1864, 1990
Foy TM, Laman JD, Ledbetter JA, Aruffo A, Ciaassen E, Noelle RJ: gp39-CD40 interactions are essential for germinal center formation and the development of B cell memory. J Exp Med 180:157–163, 1994
Foy TM, Shepherd DM, Duire FH, Aruffo A, Ledbetter JA, Noelle RJ: In vivo CD40-gp39 interactions are essential for thymus-dependent humoral immunity. II. Prolonged suppression of the humoral immune response by an antibody to the ligand for CD40, gp39. J Exp Med 178:1567–1575, 1993
Lane P, Burdet C, Hubele S, Scheidegger D, Muller U, McConnel F, Kosco-Vilbois M: B cell function in mice transgenic for m CTLA-4-Hγl: Lack of germinal centers correlated with poor affinity maturation and class switching despite normal priming of CD4+ T cells. J Exp Med 179:819–830, 1994
Linsley PS, Wallace PM, Johnson J, Gibson MG, Greene JL, Ledbetter JA, Singh C, Tepper MA: Immunosuppression in vivo by a soluble form of the CTLA-4 T cell activation molecule. Science 257:792–795, 1992
Ronchese F, Hausemann B, Hubelle S, Lane P: Mice transgenic for a soluble form of murine CTLA-4 show enhanced expansion of antigen-specific CD4+T cells and defective antibody production in vivo. J Exp Med 179:809–817, 1994
Lenschow DJ, Zeng Y, Thistlewaite JR, Montaf A, Brady W, Gibson MG, Linsley PS, Bluestone JA: Long-term survival of xenogeneic pencreatic islet grafts induced by CTLA-4Ig. Science 257:789–792, 1992
Noelle RJ, Roy M, Shepherd DM, Stamenkovic I, Ledbetter JA, Aruffo A: A 39-kDa protein on activated helper T cells binds CD40 and transduces the signal for cognate activation of B cells. Proc Natl Acad Sci USA 89:6550–6554, 1992
Klaus GGB, Humphrey JH: The generation of momery cell. I. The role of C3 in the generation of B memory cells. Immunology 33:31–40, 1977
Croix DA, Ahearn JM, Rosengard AM, Han SH, Kelsoe G, Ma M, Carroll MC: Antibody response to a T-dependent antigen requires B cell expression of complement receptors (submitted for publication)
Cosgrove D, Gray D, Dierich A, Kaufman J, Lemeur M, Benoist C, Mathis D: Mice lacking MHC class II molecules. Cell 66:1051–1066, 1991
Liu Y-J, Joshua DE, Williams GT, Smith CA, Gordon J, MacLennan ICM: Mechanism of antigen-driven selection in germinal centers. Nature 342:929–931, 1989
Han SH, Zheng B, Dal Porto J, Kelsoe G: In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl. IV. Affinity-dependent, antigen-driven B cell apoptosis in germinal centers as a mechanism for maintaining self tolerance. J Exp Med (in press) 1995
Van den Ertwegh AJM, Noelle RJ, Roy M, Shepherd DM, Aruffo A, Ledbetter JA, Boersma WJA, Classen E: In vivo CD40-gp39 interactions are essential for thymus-dependent humoral immunity. I. In vivo expression of CD40 ligand, cytokines, and antibody production. J Exp Med 178:1555–1565, 1993
Clarke SH, Huppi K, Ruezinsky D, Standt L, Gerhard W, Weigert M: Inter-and intraclonal diversity in the antibody response to influenza hemagglutinin. J Exp Med 161:687–740, 1985
Allen D, Cumano A, Dildrop R, Kocks C, Rajewsky K, Rajewsky N, Rose J, Sablitzky F, Siekevitz M: Timing, genetic requirements and somatic hypermutation during B-cell development. Immunol Rev 96:5–22, 1987
Berek C, Milstein C: The dynamic nature of the antibody repertoire. Immunol Rev 96:23–41, 1987
McKean D, Huppi K, Bell M, Staudt L, Gerhard W, Weigert M: Generation of antibody diversity in the immune response of BALB/c mice to influenza virus hemagglutinin. Proc Natl Acad Sci USA 81:3180–3184, 1984
Kepler T, Perelson A: Cyclic re-entry of germinal center B cells and the efficiency of affinity maturation. Immunol Today 14:412–415, 1993
Jacob J, Przylepa J, Miller C, Kelsoe G: In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl. III. The kinetics of V-region mutation and selection in germinal center B cells. J Exp Med 178:1293–1307, 1993
Golding GB, Gerhert PJ, Glickman BW: Patterns of somatic mutations in immunoglobulin variable genes. Genetics 115:169–176, 1987
Both GW, Taylor L, Pollard JW, Steele EJ: Distribution of mutations around rearranged heavy-chain antibody variableregion genes. Mol Cell Biol 10:5187–5196, 1990
Weber JS, Berry J, Manser T, Claflin JFL: Positions of the rearranged V kappa and its 5′ flanking sequences determines the location of somatic mutations in the J locus. J Immunol 146:3652–3655, 1991
Betz AG, Neuberger MS, Milstein C: Discriminating intrinsic and antigen-selected mutational hotspots in immunoglobulin V genes. Immunol Today 14:405–411, 1993
Rogozin IB, Kolchanov NA: Somatic hypermutagenesis in immunoglobulin genes. II. Influence of neighboring base sequences on mutagenesis. Biochim Biophys Acta 1171:11–18, 1992
Betz AG, Milstein C, Gonzalez-Fernandez A, Pannell R, Larson T, Neuberger MS: Elements regulating somatic hypermutation of an immunoglobulinκ gene: Critical role for the intron enhancer/matrix attachment region. Cell 77:239–248, 1994
Yelamos J, Klix N, Goyenechea B, Lozano F, Chui YL, Gozales-Fernandez A, Pannell R, Neuberger MS, Milstein C: Targeting of non-Ig sequences in place of the V segment by somatic hypermutation. Nature 376:225–229, 1995
Berek C, Ziegner M: The maturation of the immune response. Immunol Today 14:400–404, 1993
Kocks C, Rajewsky K: Stepwise interclonal maturation of antibody affinity through somatic hypermutation. Proc Natl Acad Sci USA 85:8206–8210, 1988
Weiss U, Rajewsky K: The repertoire of somatic antibody mutants accumulating in the memory compartment after primary immunization is restricted through affinity maturation and mirrors that expressed in the secondary response. J Exp Med 172:1681–1689, 1990
Weiss U, Zoebelein R, Rajewsky K: Accumulation of somatic mutants in the B cell compartment after primary immunization with a T cell-dependent antigen. Eur J Immunol 2:511–517, 1992
Coico RF, Bohgal BS, Thorbecke GJ: Relationship of germinal centers in lymphoid tissue to immunologic memory. VI. Transfer of B cell memory with lymph node cells fractionated according to their receptors for peanut agglutinin. J Immunol 131:2254–2258, 1983
Pascual V, Liu Y-J, Magalski A, de Bouteiller O, Banchereau J, Capra JD: Analysis of somatic mutation in five B cell subsets of human tonsil. J Exp Med 180:329–339, 1994
Weill JC, Reynaud CA: The chicken B cell compartment. Science 238:1094–1098, 1987
Pink JRL, Lassila O: B cell commitment and diversification in the bursa of Fabricius. Curr Top Microbiol Immunol 135:57–64, 1987
Fichtelius KE, Finstad J, Good RA: Bursa equivalents of bursaless vertebrates. Lab Invest 19:339–351, 1968
Cooper MD, Lawton AR: The mammalian “bursa equivalent”: Does lymphoid differentiation along plasma cell lines begin in the gut-associated lympho-epithelial (GALT) of mammals? Contemp Top Immunobiol 1:49–68, 1972
Weill JC, Reynaud CA, Lassila O, Pink JRL: Rearrangement of chicken immunoglobulin genes is not an ongoing process in the embryonic bursa of Fabricius. Proc Natl Acad Sci USA 83:3336–3340, 1986
McCormack WT, Tjoelker LW, Barth CF, Carlson LM, Petryniak B, Humphries EH, Thompson CB: Selection for B cells with productive IgL gene rearrangements occurs in the bursa of Fabricius during chicken embryonic development. Gene Dev 3:838–847, 1989
Reynaud CA, Anquez V, Grimai H, Weill JC: A hyperconversion mechanism generates the chicken light chain preimmune repertoire. Cell 48:379–388, 1987
McCormack WT, Tjoelker LW, Carlson LM, Petryniak B, Barth CF, Humphries EH, Thompson CB: Chicken IgL gene rearrangement involves deletion of a circular episome and addition of single nonrandom nucleotides to both coding segments. Cell 56:785–791, 1989
Thompson CG, Neiman PE: Somatic diversification of the chicken immunoglobulin light chain gene is limited to the rearranged variable gene segment. Cell 48:369–378, 1987
Takeda S, Masteller EL, Thompson CB, Buerstedde JM: RAG-2 expression is not essential for chicken immunoglobulin gene conversion. Proc Natl Acad Sci USA 89:4023–4027, 1992
Reynolds JD, Morris B: The evolution and involution of sheep Payer's patches in fetal and postnatal sheep. Eur J Immunol 13:627–635, 1983
Reynaud CA, McKay CR, Muller RG, Weill JC: Somatic generation of diversity in a mammalian primary lymphoid organ: the sheep ileal Peyer's patches. Cell 64:995–1005, 1991
Reynaud CA, Garcia C, Hein WR, Weill JC; Hypermutation generation the sheep immunoglobulin repertoire is an antigenindependent process. Cell 80:115–125, 1995
Reynolds JD: Evidence of extensive lymphocyte death in sheep Peyer's patches. I. A comparison of lymphocyte production and export. J Immunol 136:2005–2010, 1986
Motyka B, Reynolds JD: Apoptosis is associated with the extensive B cell death in the sheep ileal Peyer's patches and the chicken bursa of Fabricius: a possible role in B cell selection. Eur J Immunol 21:1951–1958, 1991
Becker RS, Knight KL: Somatic diversification of immunoglobulin heavy chain VDJ genes: evidence for somatic gene conversion in rabbits. Cell 63:987–997, 1990
Weinstein PD, Anderson AO, Mage RG: Rabbit IgH sequences in appendix germinal centers: VH diversification by gene conversion-like and hypermutation mechanisms. Immunity 1:647–659, 1994
Gay D, Saunders T, Camper S, Weigert M: Receptor editing: An approach by autoreactive B cells to escape tolerance. J Exp Med 177:999–1008, 1993
Tiegs SL, Russell DM, Nemazee D: Receptor editing in selfreactive bone marrow B cells. J Exp Med 177:1009–1020, 1993
Radic MZ, Erikson J, Litwin S, Weigert M: B lymphocytes may escape tolerance by revising their receptors. J Exp Med 177:1165–1173, 1993
Rolink A, Grawunder U, Haasner D, Strasser A, Melchers F: Immature surface Ig+ B cells can continue to rearrange kapper and lambda L chain gene loci. J Exp Med 178:1263–1270, 1993
Broduer PH, Osman GE, Mackle JJ, Lalor TM: The organization of the mouse Igh-V locus. Dispersion, interspersion, and the evolution of the VH gene family clusters. J Exp Med 168:2261–2278, 1988
Van Ness BG, Coleclough C, Perry RP, Weigert M: DNA between variable and joining gene segments of immunoglobulinκ light chain is frequently retained in cells that rearrange theκ locus. Proc Natl Acad Sci USA 79:262–266, 1982
Shapiro MA, Weigert M: How immunoglobulin Vκ genes rearrange. J Immunol 139:3834–3839, 1987
Clarke S, McCray S: A sharedκ reciprocal fragment and a high frequency of secondary Jκ5 rearrangements among influenza hemagglutinin specific B cell hybridomas. J Immunol 146:343–349, 1991
Miller J, Ogden S, McMullen M, Andres H, Storb U: The order and orientation of mouse A-genes explain λ-rearrangement patterns. J Immunol 141:2497–2502, 1988
Carson S, Wu GE: A linkage map of the mouse immunoglobulin lambda light chain locus. Immunogenetics 29:173–179, 1989
Harada K, Yamagishi H: Lack of feedback inhibition of Vκ gene rearrangement by productively rearranged allelea. J Exp Med 173:409–415, 1991
Feddersen RM, Van Ness BG: Double recombination of a single immunoglobulin κ-chain allele: Implications for the mechanism of rearrangement. Proc Natl Acad Sci USA 82:4793–4797, 1985
Tillman DM, Jou NT, Hill RJ, Marion TN: Both IgM and IgG anti-DNA antibodies are the products of clonally selective B cells stimulation in (NZB x NZW) F1 mice. J Exp Med 176:761–779, 1991
Makinodan T, Perkins EH, Chen MG: Immunologic activity of the aged. Adv Gerontol Res 3:171–198, 1971
Price GB, Makinodan T: Immunologic deficiencies in senescence. I. Characterization of intrinsic deficiencies. J Immunol 108:403–412, 1972
Nordin AA, Makinodan T: Humoral immunity in aging. Fed Proc 33:2033–2035, 1974
Riley SC, Froscher BG, Linton PJ, Zharhary D, Marcu K, Klinman NR: Altered VH gene segment utilization in the response to phosphorylcholine by aged mice. J Immunol 143:3798–3805, 1989
Clark SH, Claflin JL, Potter M, Rudikoff S: Polymorphisms in antiphosphorylcholine antibodies reflecting evolution of immunoglobulin families. J Exp Med 157:98–113, 1983
Perlmutter RM, Kearney JF, Chang SP, Hood LE: Developmentally controlled expression of immunoglobulin VH genes. Science 227:1597–1601, 1985
Malynn BA, Yancopoulous GD, Barth JE, Bona CA, Alt FW: Biased expression of JH-proximal VH genes occurs in the newly generated repertoire of neonatal and adult mice. J Exp Med 171:843–859, 1990
Goidl EA, Inners JB, Weksler ME: Immunological studies of aging. II. Loss of IgG and high avidity plaque-forming cells and increased suppressor cell activity in aging mice. J Exp Med 144:1037–1048, 1976
Kishimoto S, Takahama T, Mizumachi H: In vitro immune response to the 2,4,6-trinitrophenyl determinant in aged C57BL/6J mice: changes in the humoral immune response to, avidity for the TNP determinant and responsiveness to LPS effect with aging. J Immunol 116:294–300, 1976
Doria G, D'Agostaro G, Poretti A: Age-dependent variations in antibody avidity. Immunology 35:601–611, 1978
Miller C, Kelsoe G, Han SH: Lack of B7-2 expression in the germinal centers of aged mice. Aging Immun Inf Dis 5:249–257, 1994
Miller C, Kelsoe G: Somatic hypermutation is absent in aged mice. J Immunol (in press), 1995
Szakal AK, Taylor JK, Smith JP, Kosco MH, Burton GF, Tew JG: Kinetics of germinal center development in lymph nodes of young and aging immune mice. Anat Rec 227:475, 1990
Engwerda CR, Handwerger BS, Fox BS: Aged T cells are hyporesponsive to costimulation mediated by CD28. J Immunol 152:3470–3477, 1994
Korthauer U, Graf D, Mages HW, Briere F, Padayachee M, Malcolm S, Ugazio AG, Notarangelo LD, Levinsky RJ, Kroczek RA: Defective expression of T cell CD40 ligand cause X-linked immunodeficiency with hyper-IgM. Nature 361:539–541, 1993
DiSanto JP, Bonnefoy JY, Gauchat JF, Fischer A de Saint Basile G: CD40 ligand mutations in X-linked immunodeficiency with hyper-IgM. Nature 361:541–543, 1993
Vetrie D, Vorechovsky I, Sideras P, Holland J, Davies A, Flinter F, Hammarstrom L, Kinnon C, Levinsky R, Bobrow M, Smith CI, Bentley DR: The gene involved in X-linked agammaglobulinaemia is a member of the src family of protein-tyrosine kinases. Nature 361:226–233, 1993
Tsukada S, Saffran DC, Rawlings DJ, Parolini O, Allen RC, Klisak I, Sparkes RS, Kubagawa H, Thuluvancheri M, Quan S, Belmont JW, Cooper MD, Conley ME, Witte ON: Deficient expression of a B cell cytoplasmic tyrosine kinase in human X-linked agammaglobulinemia. Cell 72:271–290, 1993
Noguchi M, Huafang Y, Rosenblatt HM, Filipovitch AH, Adelstein S, Modi WS, McBride OW, Leonard WJ: Interleukin-2 receptorγ chain mutation results in X-linked severe combined immunodeficiency in human. Cell 73:147–157, 1993
Derry JM, Ochs HD, Francke U: Isolation of a novel gene mutated in Wiskott-Aldrich syndrome. Cell 78:635–644, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zheng, B., Kelsoe, G. & Han, S. Somatic diversification of antibody responses. J Clin Immunol 16, 1–11 (1996). https://doi.org/10.1007/BF01540967
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01540967