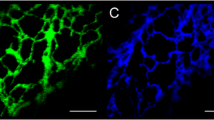
Summary
Recent fluorescence, AVEC-DIC, and confocal laser scanning microscopic studies have revealed the dynamic nature and structural extent of a calcium-sequestering endoplasmic reticulum (ER) in plant cells. Various investigators have proposed different roles for the ER in cell motility. One, the ER plays a direct role in the generation of intracellular particle motions or two, the ER regulates particle motions indirectly. We show that the ER can be extruded fromAcetabularia cells, stains brightly with the fluorescent dye DiOC6(3), and small (ca. 100 nm diameter) fluorescent vesicles are observed to move in or along the ER tubules. Intracellular particle movements in the giant algal cellAcetabularia can be transiently inhibited by IP4, IP3, and IP2, compounds which in animal cells are known to cause the release of free calcium ions. A model is proposed which clarifies the possible relationships between the ER, calciosomes, calcium ions, and the microfilament-generated intracellular particle movements observed in plant cells.
Similar content being viewed by others
Abbreviations
- AVEC-DIC:
-
video microscopy in differential interference contrast
- CFLSM:
-
confocal laser scanning microscope
- DiOC6(3):
-
3,3′-dihexyloxacarbocyanine iodide
- ER:
-
endoplasmic reticulum
- IP3 :
-
inositol triphosphate
- N.A.:
-
numerical aperture
- SIT:
-
silicon intensified target video camera
- SR:
-
sarcoplasmic reticulum
References
Allen NS (1980) Cytoplasmic streaming and transport in the characean algaNitella. Can J Bot 58: 786–796
—, Brown DT (1988) Dynamics of the endoplasmic reticulum in living onion epidermal cells in relation to microtubules, microfilaments, and intracellular particle movement. Cell Motil Cytoskeleton 10: 153–163
—, O'Connor SA (1990) Inhibition of intracellular particle motions inAcetabularia acetabulum L. by phosphoinositides. In: Morré DJ, Boss WF, Loewus FA (eds) Plant inositides. Alan R Liss, New York, pp 301–310
Allen RD, Allen NS, Travis J (1981) Video-enhanced contrast, differential interference contrast (AVEC-DIC) microscopy: a new method capable of analyzing microtubule-related motility in the reticulopodial network ofAllegromia laticollaris. Cell Motil 1: 291–302
Berridge MJ (1987) Inositol trisphosphate and diacylglycerol: two interacting second messengers. Annu Rev Biochem 56: 159–194
Chou MK, Krause K, Campbell K, Jensen K, Sjolund R (1989) Calsequestrin: a novel calcium binding protein in plants. Plant Physiol 89: 149
Fliegel L, Ohnishi M, Carpenter MR, Khanna VK, Reithmeier RAF, McLennan DH (1987) Amino acid sequence of rabbit fast-twitch skeletal muscle calsequestrin deduced from cDNA and peptide sequencing. Proc Natl Acad Sci USA 84: 1167–1171
Hashimoto S, Bruno B, Lew DP, Pozzan T, Volpe P, Meldolesi J (1988) Immunocytochemistry of calciosomes in liver and pancreas. J Cell Biol 107: 2523–2531
Hayama T, Shimmen T, Tazawa M (1979) Partizipation of Ca++ in cessation of cytoplasmic streaming induced by membrane excitation in characeae internodal cells. Protoplasma 99: 305–321
Kachar B, Reese TS (1988) The mechanism of cytoplasmic streaming in characean algal cells: sliding of endoplasmic reticulum along actin filaments. J Cell Biol 106: 1545–1552
Krause KM, Chou M, Thomas MA, Sjolund RD (1989) Plant cells contain calsequestrin. J Biol Chem 264: 4269–4272
Marmé D (1989) The role of calcium and calmodulin in signal transduction. In: Boss WF, Morré DJ (eds) Second messengers in plant growth and development. Alan R Liss, New York, pp 57–80
Nagai R, Hayama T (1979) Ultrastructure of the endoplasmic factor responsible for cytoplasmic streaming inChara internodal cells. J Cell Sci 36: 121–136
Oberdorf JA, Head JF, Kaminer B (1986) Calcium uptake and release by isolated cortices and microsomes from the unfertilized egg of the sea urchinStrongylocentrotus droebachiensis. J Cell Biol 102: 2205–2210
—, Lebeche D, Head JF, Kaminer B (1988) Identification of a Calsequestrin-like protein from sea urchin eggs. J Biol Chem 263: 6806–6809
Palade GE, Porter KR (1954) Studies on the endoplasmic reticulum. I. Its identification in cells in situ. J Exp Med 100: 541–656
Quader H, Hoffmann A, Schnepf E (1987) Shape and movement of the endoplasmic reticulum in onion bulb epidermis cells: possible involvement of actin. Eur J Cell Biol 44: 17–26
Scott BT, Simmerman HKB, Collins JH, Nadal-Ginard B, Jones LR (1988) Complete amino acid sequence of canine cardiac calsequestrin deduced by cDNA cloning. J Biol Chem 263: 8958–8964
Sheetz MP, Spudich JP (1983) Movement of myosin-coated fluorescent beads on actin cables in vitro. Nature 303: 31–35
Teresaki M, Serdat C (1989) Localization of calcium uptake and release to sea urchin egg cortical ER using the calcium indicator dye Fluo-3. J Cell Biol 109: 302 a
—, Chen LV, Fujiwara K (1986) Microtubules and the endoplasmic reticulum are highly interdependent structures. J Cell Biol 103: 1557–1568
Volpe P, Krause KH, Hashimoto S, Zorzato F, Pozzan T, Meldolesi J, Lew DP (1988) “Calciosome”, a cytoplasmic organelle: the inositol-l,4,5-triphosphate-sensitive Ca++ store of nonmuscle cells? Proc Natl Acad Sci USA 85: 1091–1095
Williamson RE (1979) Filaments associated with the endoplasmic reticulum in the streaming cytoplasm ofChara corallina. Eur J Cell Biol 20: 177–183
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Strömgren Allen, N., Schumm, J.H. Endoplasmic reticulum, calciosomes and their possible roles in signal transduction. Protoplasma 154, 172–178 (1990). https://doi.org/10.1007/BF01539845
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01539845