Abstract
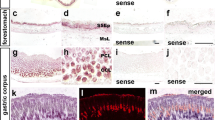
The expression and distribution of deoxyribonuclease I (DNase I) in rat parotid gland, kidney, small intestine and keratinized epithelium was further analysed at the level of its mRNA by in situ hybridization and correlated to immunohistochemical results using polyclonal anti-DNase I antibodies. High amounts of DNase I-specific mRNA and immunoreactivity were detected in the parotid gland, kidney and small intestine in agreement with previous immunohistochemical results. In the parotid gland, both the DNase I-specific mRNA and antigenicity were detected within the secretory cells. In the kidney, DNase I gene transcripts were localized in distal tubules and the collecting duct system. In this organ an identical localization of DNase I antigenicity was obtained. In the small intestine only the enterocytes covering the villi were shown to express DNase I-specific mRNA; the highest level being detected within the enterocytes along the lower third of the villi. In contrast, the highest level of immunoreactivity was found in enterocytes covering the middle and upper thirds of the villi. Within the stratified epithelium of the tongue, DNase I gene transcription and protein expression started in lower parts of the stratum spinosum and reached into the stratum granulosum. However, the gradient of DNase I gene transcript expression appeared to be shifted to lower layers of the stratum spinosum when compared to DNase I immunoreactivity. No Dnase I expression was found in the keratinocytes of the basal cell layer. The results obtained for the small intestine and stratified epithelium allowed a temporal analysis of the time course of DNase I expression at the level of mRNA and protein: DNase I-specific mRNA peaked before protein biosynthesis. In contrast, apoptotic elimination of the terminally differentiated entero- and keratinocytes was shown to occur only at the uppermost villar tips or underneath the keratinized layer, as demonstrated by in situ end-labelling of free 3′-OH ends of cleaved DNA.
Similar content being viewed by others
References
Barry MA, Eastman A (1993) Identification of deoxyribonuclease II as an endonuclease involved in apoptosis. Arch Biochem Biophys 300:440–450
Caron-Leslie L-AM, Schwartzman RA, Gaido ML, Crompton MM, Cidlowski JA (1991) Identification and characterization of glucocorticoid-regulated nuclease(s) in lymphoid cells undergoing apoptosis. J Steroid Biochem Biol 40:661–671
Gaido ML, Cidlowski JA (1991) Identification, purification, and characterization of a calcium-dependent endonuclease (NUC18) from apoptotic rat thymocytes. J Biol Chem 266:18580–18585
Gavrieli Y, Sherman Y, Ben-Sasson SA (1992) Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol 119:493–501
Hewish DR, Burgoyne LA (1973) Chromatin sub-structure. The digestion of chromatin DNA at regularly spaced sites by a nuclear deoxyribonuclease. Biochem Biophys Res Commun 52:504–542
Kishi K, Yasuda T, Ikehara Y, Sawazaki K, Sato W, Iida R (1990) Human serum deoxyribonuclease I (DNase I) polymorphism: pattern similarities among isoenzymes from serum, urine, kidney, liver, and pancreas. Am J Hum Genet 47:121–126
Kreuder V, Dieckhoff J, Sittig M, Mannherz HG (1984) Isolation, characterisation and crystallisation of deoxyribonuclease I from bovine and rat parotid gland and its interaction with rabbit skeletal muscle actin. Eur J Biochem 139:389–400
Lacks SA (1981) Deoxyribonuclease I in mammalian tissues. Specificity of inhibition by actin. J Biol Chem 256:2644–2648
Malicka-Blaszkiewicz M, Roth JS (1983) Evidence for the presence of DNase-actin complex in L1210 leukemia cells. FEBS Lett 153:235–239
Mannherz HG, Kreuder V, Koch J, Dieckhoff J, Drenckhahn D (1982) The inhibition of bovine and rat deoxyribonuclease I by skeletal muscle actin. A biochemical and immunocytological study. Biochem J 207:305–307
Oberhammer FA, Pavelka M, Sharma S, Tiefenbacher R, Purchino AF, Bursch W, Schulte-Hermann R (1992) Induction of apoptosis in cultured hepatocytes and in regressing liver by transforming growth factor β1. Proc Natl Acad Sci USA 89:5408–5412
Peitsch MC, Hesterkamp T, Polzar B, Mannherz HG, Tschopp J (1992) Functional characterization of serum DNase I in MRL-lpr/lpr mice. Biochem Biophys Res Commun 186:739–745
Peitsch MC, Polzar B, Stephan H, Crompton T, MacDonald HR, Mannherz HG, Tschopp J (1993) Characterization of the endogenous deoxyribonuclease involved in nuclear DNA degradation during apoptosis (programmed cell death). EMBO J 12:371–377
Peitsch MC, Mannherz HG, Tschopp J (1994) The apoptosis endonucleases: cleaning up after cell death. Trends Cell Biol 4:37–41
Polzar B, Mannherz HG (1990) Nucleotide sequence of a full length cDNA clone encoding the deoxyribonuclease I from rat parotid gland. Nucleic Acids Res 18:7151
Polzar B, Rösch A, Mannherz HG (1989) A simple procedure to prepare monospecific polyclonal antibodies of high affinity against actin from muscular sources. Eur J Cell Biol 50:220–229
Polzar B, Peitsch MC, Loos R, Tschopp J, Mannherz HG (1993) Overexpression of deoxyribonuclease I (DNase I) transfected into COS-cells: its distribution during apoptotic cell death. Eur J Cell Biol 62:397–405
Polzar B, Zanotti S, Stephan H, Rauch F, Peitsch MC, Irmler M, Tschopp J, Mannherz HG (1994) Distribution of deoxyribonuclease I in rat tissues and its correlation to cellular turnover and apoptosis (programmed cell death). Eur J Cell Biol 64:200–210
Potten CS (1982) The significance of spontaneous and induced apoptosis in the gastrointestinal tract of mice. Cancer Metastasis Rev 11:179–195
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Schumer M, Colombel MC, Sawcuk IS, Gobé G, Connor J, Toole KM-O, Olsson CA, Wise GJ, Buttyan R (1992) Morphologic, biochemical, and molecular evidence of apoptosis during the reperfusion phase after brief periods of renal ischemia. Am J Pathol 140:831–838
Studier FW, Moffat BA (1986) Use of bacterophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J Mol Biol 189:113–130
Ucker DS, Obermiller PS, Eckhart W, Apgar JR, Bergwer NA, Meyers J (1992) Genome digestion is a dispensable consequence of physiological cell death mediated by cytotoxic T lymphocytes. Mol Cell Biol 12:3060–3069
Wijsman JH, Jonker RR, Keijzer R, van de Velde CJH, Cornelisse CJ, van Dierendonck JH (1993) A new method to detect apoptosis in paraffin sections: in situ end-labeling of fragmented DNA. J Histochem Cytochem 41:7–12
Wyllie AH (1980) Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 284:555–556
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zanotti, S., Polzar, B., Stephan, H. et al. Localization of deoxyribonuclease I gene transcripts and protein in rat tissues and its correlation with apoptotic cell elimination. Histochem Cell Biol 103, 369–377 (1995). https://doi.org/10.1007/BF01457812
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01457812