Abstract
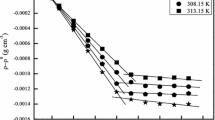
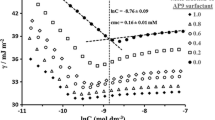
The phase equilibrium between the two isotropic phases formed above the cloud point in an aqueous solution containing anionic surfactant, nonionic surfactant, and salt was studied as a function of the anionic/total surfactant ratio in the concentrated or coacervate phase under isothermal conditions. As the anionic/total surfactant ratio increased, the system approached the lower consolute solution temperature and the two phases became increasingly similar in composition. The concentration of surfactant in the dilute phase was found to be well above the CMC. The activity coefficient of each component was determined in the coacervate phase. The activity coefficients of the surfactant components in the coacervate were also computed on a surfactant-only basis. There is substantial negative deviation from ideality of mixing between the dissimilar surfactants in the coacervate. Regular solution theory provides an approximate description of the anionic/nonionic surfactant interaction in the coacervate. A thermodynamic consistency test found the true activity coefficients in the coacervate to be internally consistent, but the surfactant-only based activity coefficients failed the test, bringing into question the validity of using the pseudo-phase separation model to describe surfactant interactions in the coacervate.
Similar content being viewed by others
Abbreviations
- a A :
-
anionic surfactant activity
- a i :
-
activity of componenti
- a N :
-
nonionic surfactant activity
- a S :
-
activity of salt
- a W :
-
activity of water
- C A :
-
anionic surfactant monomer concentration
- C 0A :
-
anionic surfactant standard state concentration
- C TA :
-
total anionic surfactant concentration in dilute phase
- C M :
-
total surfactant concentration present as micelles in dilute phase
- C N :
-
nonionic surfactant monomer concentration
- C 0N :
-
nonionic surfactant standard state concentration
- C TN :
-
total nonionic surfactant concentration in dilute phase
- CMC :
-
critical micelle concentration
- CMC A :
-
anionic surfactant critical micelle concentration
- CMC M :
-
anionic/nonionic surfactant mixture critical micelle concentration
- CMC N :
-
nonionic surfactant critical micelle concentration
- R :
-
ideal gas constant
- T :
-
absolute temperature
- W C :
-
interaction parameter in the coacervate
- W M :
-
micellar interaction parameter
- X A :
-
anionic surfactant-only based mole fraction in the micelle
- X N :
-
nonionic surfactant-only based mole fraction in the micelle
- Y A :
-
anionic surfactant-only based monomer mole fraction
- Y N :
-
nonionic surfactant-only based monomer mole fraction
- Z A :
-
anionic surfactant mole fraction in coacervate
- Z A, S :
-
anionic surfactant-only based mole fraction in the coacervate
- Z i :
-
mole fraction of componenti in coacervate
- Z N :
-
nonionic surfactant mole fraction in coacervate
- Z N, S :
-
nonionic surfactant-only based mole fraction in the coacervate
- Z S :
-
mole fraction of salt in coacervate
- Z W :
-
mole fraction of water in coacervate
- γ A :
-
anionic surfactant activity coefficient in coacervate
- γ A, M :
-
anionic surfactant activity coefficient in the micelle
- γ A, S :
-
anionic surfactant-only based activity coefficient in the coacervate
- γ i :
-
activity coefficient of componenti in coacervate
- γ n :
-
nonionic surfactant activity coefficient in coacervate
- γ N, M :
-
nonionic surfactant activity coefficient in the micelle
- γ N, S :
-
nonionic surfactant-only based activity coefficient in the coacervate
- γ S :
-
activity coefficient of salt in coacervate
- γ W :
-
activity coefficient of water in coacervate
References
Smith DH, Fleming PD (1985) J Colloid Interface Sci 105:80
Scamehorn JF, Schechter RS, Wade WH (1982) J Dispersion Sci Technol 3:261
Rosen MJ, Hua XY (1982) J Am Oil Chem Soc 59:582
Holland PM, Adv Colloid Interface Sci, in press
Rubingh DN (1979) (ed) Mittal KL, in Solution Chemistry of Surfactants, Plenum Press, New York p 337
Scamehorn JF, Schechter RS, Wade WH (1982) J Colloid Interface Sci 85:494
Lunkenheimer K, Wantke KD (1981) Colloid Polymer Sci 259:354
Saito H, Shinoda K (1967) J Colloid Interface Sci 24:10
Maclay WN (1956) J Colloid Sci 11:272
Nakagawa T, Shinoda K (1963) (eds) Shinoda K, Tamamushi B, Nakagawa T, Isemura T, in Colloidal Surfactants, Academic Press, New York p 130
Nishikido N, Akisada H, Matuura R (1977) Mem Fac Sci Kyushu Univ, Ser C 10:91
Kuriyama K, Inoue H, Nakagawa T (1962) Kolloid-Z 183:68
Tadros ThF (1974) J Colloid Interface Sci 46:528
Pletnev MYu, Trapeznikov AA (1978) Kolloidn Zh 40:1126
Corti M, Minero C, Degiorgio V (1984) J Phys Chem 88:309
Rathman JF, Scamehorn JF (1984) J Phys Chem 88:5807
Tsumori T, Nishikido N, Moroi Y, Matuura R (1974) Mem Fac Sci Kyushu Univ, Ser C 9:57
Nishikido N, Yoshimura N, Tanaka M (1980) J Colloid Interface Sci 78:338
Mitchell DJ, Tiddy GJT, Waring L, Bostock T, McDonald MP (1983) J Chem Soc Faraday Trans I 79:975
Lindman B, Wennerstrom H (1980) (ed) Boschke FL, in Topics in Current Chemistry, Springer-Verlag, West Berlin Vol 87 p 1
Kjellander R (1982) J Chem Soc Faraday Trans II 78:2025
Nilsson PG, Lindman B (1983) J Phys Chem 87:4756
Lang JC, Morgan RD (1980) J Chem Phys 73:5849
Doren J, Goldfarb J (1970) J Colloid Interface Sci 32:67
Corti M, Degiorgio V, Zulauf M (1982) Phys Rev Lett 48:1617
Wan LSC (1983) J Am Oil Chem Soc 60:1359
Sepulveda L, MacRitchie F (1968) J Colloid Interface Sci 28:19
Balzhizer RE, Samuels MR, Eliassen JD (1972) Chemical Engineering Thermodynamics, Prentice-Hall Englewood Cliffs, New Jersey Ch 10
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoesting, O.E., Scamehorn, J.F. Phase equilibrium in aqueous mixtures of nonionic and anionic surfactants above the cloud point. Colloid & Polymer Sci 264, 148–158 (1986). https://doi.org/10.1007/BF01414842
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01414842