Summary
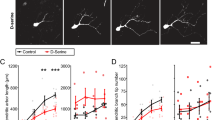
The amino acid taurine plays an important trophic role during development and regeneration of the central nervous system. Other amino acid systems, such as those for glutamate and gamma-aminobutyric acid (GABA), are modified during the same physiological and pathological processes. After crushing the optic nerve, goldfish retinal explants were plated in the absence and in the presence of different amino acids and amino acid receptor agonists. The length and the density of the neurites were measured at 5 days in culture. Taurine increased the length and the density of neurites. Glutamate and glycine increased them at low concentration, but were inhibitors at higher concentration. The combination of N-methyl-D-aspartate (NMDA) and glycine produced a greater inhibitory effect than NMDA alone. NMDA or alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) added simultaneously with taurine impaired the stimulatory effect of the latter. GABA stimulated the emission of neurites in a concentration dependent manner. Hypotaurine also elevated the length of neurites, but cysteinesulfinic acid did not produce a significant effect. The concentrations of taurine, glutamate and GABA were determined by HPLC with fluorescent detection in the retina of goldfish at various days post-crushing the optic nerve. The levels of taurine were significantly increased at 48 h after the crush, and were elevated up to 20 days. Glutamate level decreased after the lesion of the optic nerve and was still low at 20 days. GABA concentration was not significantly different from the control. The interaction of these amino acids during the regenerative period, especially the balance between taurine and glutamate, may be a determinant in restoring vision after the crush.
Similar content being viewed by others
Abbreviations
- AMPA:
-
alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid
- GABA:
-
gamma-aminobutyric acid
- NMDA:
-
N-methyl-D-aspartate
References
Akimoto J, Itoh H, Miwa T, Ikeda K (1993) Immunohistochemical study of glutamine synthetase expression in early glial development. Brain Res Dev Brain Res 72: 9–14
Araki M, Tone S, Akagawa K, Kimura H (1995) High potassium promotes differentiation of retinal neurons but does not favor rod differentiation. Brain Res Dev Brain Res 89: 103–114
Bagley PJ, Hirschberger LL, Stipanuk MH (1995) Evaluation and modification of an assay procedure for cysteine dioxygenase activity: high-performance liquid chromatography method for measurement of cysteine sulfinate and demonstration of physiological relevance of cysteine dioxygenase activity in cysteine catabolism. Anal Chem 227: 40–48
Balázs R, Hack N, Jorgensen OS (1990a) Interactive effects involving different classes of excitatory amino acid receptors and the survival of cerebellar granule cells in culture. Int J Dev Neurosci 8: 347–359
Balázs R, Hack N, Jorgensen OS (1990b) Selective stimulation of excitatory a mino acid receptor subtypes and the survival of cerebellar granule cells in culture: effect of kainic acid. Neuroscience 37: 251–258
Barbin G, Pollard H, Gaiarsa JL, Ben-Ari Y (1993) Involvement of GABAA receptors in the outgrowth of cultured hippocampal neurons. Neurosci Lett 152: 150–154
Barlow RB (1983) Biodata handling with microcomputers. Elsevier Science Publishers, Amsterdam, pp 125–133
Behar T, Ma W, Hudson L, Barker JL (1994) Analysis of the anatomical distribution of GAD67 mRNA encoding truncated glutamic acid decarboxylase proteins in the embryonic rat brain. Brain Res Dev Brain Res 77: 77–87
Berger T, Walz W, Kettenman H (1992) GABA- and glutamate-activated currents in glial cells of the mouse corpus callosum slice. J Neurosci Res 31: 21–27
Boje KM, Skolnick P (1992) Ontogeny of glycine-enhanced [3H]MK-801 binding to N-methyl-D-aspartate receptor-coupled ion channels. Brain Res Dev Brain Res 65: 51–56
Brenneman DE, Forsythe ID, Nicol T, Nelson PG (1990) Methyl-D-aspartate receptors influence neuronal survival in developing spinal cord cultures. Brain Res Dev Brain Res 51: 63–68
Chiba C, Saito T (1995) Developmental of responses to excitatory and inhibitory amino acids in spiking cells during retinal regeneration in the adult next. Jpn J Physiol 45: 869–887
Choi DW, Maulucci-Gedde MA, Kriegstein AR (1997) Ionic dependence of glutamate neurotoxicity. J Neurosci 7: 369–379
Cobas A, Fairén C, Alvarez-Bolado G, Sánchez MP (1991) Prenatal development of the intrinsic neurons of the rat neocortex: a comparative study of the distribution of GABA-immunoreactive cells and the GABAA receptor. Neuroscience 40: 375–397
Constantine-Paton M, Cline HT, Debski E (1990) Patterned activity, synaptic convergence, and the NMDA receptors in developing visual pathways. Annu Rev Neurosci 13: 129–154
Darvas Z, Swydan R, Csaba G (1985) Influence of benzodiazepine (diazepam) by single treatment on the growth of Tetrahymena. Biomed Biochim Acta 44: S1725–1728
de la Rosa J, Stipanuk MH (1985) Evidence for a rate-limiting role of cysteinsulfinate decarbosylase activity in taurine biosynthesisin vivo. Comp Biochem Physiol 81B: 565–571
Fix AS, Horn JW, Hall RL, Johnson JA, Tizzano JP (1995) Progressive retinal toxicity in neonatal rats treated with D,L-2,amino-3-phosphonopropionate (D,L-AP3). Vet Pathol 32: 521–531
Hahn JS, Aizenman E, Lipton SA (1988) Central mammalian neurons normally resistant to glutamate toxicity are made sensitive by elevate extracellular Ca+2: Toxicity is blocked by the N-methyl-D-aspartate antagonist MK-801. Proc Natl Acad Sci USA 85:6556–6560
Hayes KC, Carey R, Schmidt S (1975) Retinal degeneration associated with taurine deficiency in the cat. Science 188: 949–951
Hieber VC, Goldman D (1995) Trans-synaptic regulation of NMDA receptor RNAs during optic nerve regeneration. J Neurosci 15: 5286–8296
Ishikawa A, Shiono T, Ishiguro S, Tamai M (1996) Postnatal development expression of glutamine and related amino acids in the rat retinas. Curr Eye Res 15: 662–668
Johnston MV (1994) Developmental aspects of NMDA receptor agonists and antagonists in the central nervous system. Psychopharmacol Bull 30: 567–575
Kater SB, Mattson MP, Cohan C, Connor J (1988) Calcium regulation of the neuronal grosth cone. Trends Neurosci 11: 315–321
Kiryu S, Yao GL, Morita N, Kato H, Kiyama H (1995) Nerve injury enhances neuronal glutamate transporter expression: identification by differential display PCR. J Neurosci 15: 7827–7878
Lake N (1988) Taurine depletion leads to loss of rat optic nerve axons. Vision Res 28: 1071–1076
Lauder JM, Han VKM, Henderson P, Verdoorn T, Towle AC (1986) Prenatal ontogeny of the GABAergic system in the rat brain: an immunocytochemical study. Neuroscience 19: 465–493
Lauder JM (1993) Neurotransmitters as growth regulatory signals: role of receptors and second messengers. Trends Pharmacol Sci 16: 233–240
Levi G, Patrizio M (1992) Astrocyte heterogeneity: endogenous amino acid levels and release evoked by non-N-methyl-D-aspartate receptor agonists and by potassium-induced swelling in type-1 and type-2 astrocytes. J Neurochem 58: 1943–1952
Lima L, Matus P, Drujan B (1988) Taurine effect on neuritic growth from goldfish retinal explants. Int J Dev Neurosci 6: 417–424
Lima L, Matus P, Drujan B (1989a) Spatial distribution of glutamate, taurine and GABA in teleosts and mammals retina:in vivo andin vitro study. Int J Dev Neurosci 7: 295–300
Lima L, Matus P, Drujan B (1989b) The interaction of substrate and taurine modulates the outgrowth from goldfish retinal explants. Int J Dev Neurosci 7: 375–382
Lima L, Matus P, Drujan B (1992) The trophic role of taurine in the retina. A possible mechanism of action. In: Lombardini JB, Schaffer SW, Azuma J (eds) Taurine nutritional value and mechanism of action. Plenum Press, New York, pp 287–294
Lima L, Matus P, Drujan B (1993) Taurine-induced regeneration of goldfish retina in culture may involve a calcium-mediated mechanism. J Neurochem 60: 2153–2158
Lipton SA, Kater SB (1989) Neurotransmitter regulation of neuronal outgrowth plasticity and survival. Trends Neurosci 12: 265–270
Lodge D, Collingridge G (1991) The pharmacology of excitatory amino acids: a TIPS special report. Trends Pharmacol Sci (special issue): 1–89
Lowry OH, Rosebrough, NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
McDonald JW, Johnston MV (1990) Physiological and pathophysiological roles of excitatory amino acids during central nervous system development. Brain Res Rev 15: 41–70
Mattson MP, Dou P, Kater SB (1988a) Outgrowth-regulating actions of glutamate in isolated hippocampal pyramidal neurons. J Neurosci 8: 2087–2100
Mattson MP, Lee RE, Adams ME, Guthrie PB, Kater SB (1988b) Interaction between entorhinal axons and target hippocampal neurons: a role for glutamate in the development of hippocampal circuitry. Neuron 1: 865–876
Mattson MP, Hauser KF (1991) Spatial and temporal integration of neurotransmitter signals in the development of neural circuitry. Neurochem Int 19: 17–24
Matus P, Obregón F, Lima L (1997) Differential effect of taurine and serotonin on the outgrowth from explants or isolated cells of the retina. Int J Dev Neurosci (in press)
Meier E, Hertz L, Schousboe A (1991) Neurotransmitters as developmental signals. Neurochem Int 19: 1–15
Messersmith EK, Redburn DA (1990) Kainic acid lesioning alters development of the outer plexiform layer in neonatal retina. Int J Dev Neurosci 8: 447–461
Nichol KA, Schulz MW, Bennett MR (1995) Nitric oxide-mediated death of cultured neonatal retinal ganglion cells: neuroprotective properties of glutamate and chondroitin sulfate proteoglycan. Brain Res 697: 1–16
Obregón F, Lima L (1996) Cysteinsulfinic acid decarboxylase in goldfish and rat retina. Acta Cient Vlna 47: 193
Pasantes-Morales H, Cruz C (1983) Possible mechanisms involved in the protective action of taurine on photoreceptor structure. In: Kuriyama K, Huxtable RJ, Iwata H (eds) Sulfur amino acids: biochemical and clinical aspects. Alan R. Liss, New York, pp 263–276
Patterson PH (1988) On the importance of being inhibited, or saying no to growth cones. Neuron 1: 263–267
Pearce IA, Cambray-Deakin MA, Burgoyne RD (1987) Glutamate acting on NMDA receptors stimulates lates neurite outgrowth from cerebellar granule cells. FEBS Lett 223: 143–147
Puel JL, Saffiedine S, Gervais-d'Aldin C, Eybalin M, Pujol R (1985) Synaptic regeneration and functional recovery after excitotoxic injury in the guinea pig cochlea. C R Acad Sci 318: 67–75
Rothman SM, Thurston JH, Hauhart RE (1987) Delayed neurotoxicity of excitatory amino acidsin vitro. Neurosci 22: 471–480
Schwartz JP (1990) Neurotransmitters as neurotrophic factors: a new set of functions. Int Rev Neurobiol 34: 1–23
Szabo G, Katarova Z, Greenspan R (1994) Distinct protein forms are produced from alternatively spliced bicistronic glutamic acid decarboxylase mRNAs during development. Moll Cell Biol 14: 7535–7545
Schmidt S, Berson EL, Hayes KC (1976) Retinal degeneration in cats fed casein. 1: Taurine deficiency. Invest Ophtalmol 15: 47–52
Sturman JA, Moretz RC, French JH, Wisnewski HM (1985a) Taurine deficiency in the developing cat: persistance of the cerebellar external granule cell layer. J Neurosci Res 13: 405–416
Sturman JA, Moretz RC, French JH, Wisnewski HM (1985b) Postnatal taurine deficiency in the kitten results in a persistance of the cerebellar external granule cell layer: correction by taurine feeding. J Neurosci Res 13: 521–528
Ueda H, Hieber V (1995) Down-regulation of AMPA-like glutamate receptor gene expression during goldfish optic nerve regeneration. Brain Res Mol Brain Res 32: 151–155
Vago JM, Marshall JF (1996) Unilateral frontal cortex ablation producing neglect causes time-dependent changes in striatal glutamate receptors. Behav Brain Res 77: 189–199
Versaux-Botteri C, Hergueta S, Pieau C, Wasowicz M, Dalil-Thiney N, Nguyen-Legros J (1994) Early development of GABA-like immunoreactive cells in the retina of turtle embryos. Brain Res Dev Brain Res 83: 125–131
Wong RO (1995) Effects of glutamate and its analogs on intracellular calcium levels in the developing retina. Vis Neurosci 12: 907–917
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lima, L., Obregón, F. & Matus, P. Taurine, glutamate and GABA modulate the outgrowth from goldfish retinal explants and its concentrations are affected by the crush of the optic nerve. Amino Acids 15, 195–209 (1998). https://doi.org/10.1007/BF01318859
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01318859