Summary
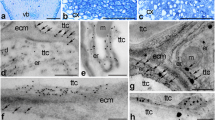
Cellulase activity was localized at the ultrastructural level in pollen mother cells (PMCs) of David lily [Lilium davidii var.willmottiae (Wilson) Roffill] at different stages of meiotic prophase I. The enzyme was observed to appear at the early leptotene stage and reached its highest level at the subsequent zygotene stage, and its subcellular distribution revealed by the presence of electron-dense deposits of reaction product was found to be restricted exclusively to the endoplasmic reticulum (ER), the vesicles derived from that, and the cell wall, especially at the sites of secondary plasmodesmata and cytoplasmic channels where the wall was being digested. Other cytoplasmic organelles, such as dictyosomes and Golgi vesicles, lacked such deposits of reaction product. After zygotene the enzyme activity decreased abruptly, and at the pachytene stage only very few deposits could be observed in the cell wall. Our results indicate that cellulase is synthesized on rough ER and secreted directly via the smooth ER and ER-derived vesicles into the cell wall by exocytosis, where it brings about local wall breakdown, leading to the secondary formation of plasmodesmata and cytoplasmic channels.
Similar content being viewed by others
References
Bal AK (1972) Localization of cellulase in plant cells. In: Takeuchi T, Agawa K, Frigita S (eds) Proceedings of the 4th International Congress for Histochemistry and Cytochemistry, Kyoto, Japan, pp 301–302
— (1974) Cellulase. In: Hayat MA (ed) Electron microscopy of enzymes, vol 3. Van Nostrand Reinhold, New York, pp 68–76
—, Verma DPS, Byrne H, Maclachlan GA (1976) Subcellular localization of cellulase in auxin-treated pea. J Cell Biol 69: 97–105
Binding H, Witt D, Monzer J, Mordhorst, Kollmann R (1987) Plant cell graft chimeras obtained by co-culture of isolated protoplasts. Protoplasma 141: 64–73
Brummell DA, Catala C, Lashbrook CC, Bennett AB (1997) A membrane-anchored E-type endo-1,4-glucanase is localized on Golgi and plasma membranes of higher plants. Proc Natl Acad Sci USA 94: 4794–4799
Carr DJ (1976) Plasmodesmata in growth and development. In: Gunning BES, Robards AW (eds) Intercellular communication in plants: studies on plasmodesmata. Springer, Berlin Heidelberg New York, pp 243–249
Cheng KC (1956) On the process of intercellular migration of chromatin substance and new formation of nucleus in the pollen mother cells ofLillium sutchueneuse Franch. Sci Sin 5: 497–507
—, Nie SW, Chen SW, Jian LC, Sun LH, Sun DL (1987) Studies on the secondary formation of plasmodesmata between the pollen mother cells of lily before cytomixis. Acta Biol Exp Sin 20: 1–11
Ding B, Haudenshield JS, Hull RJ, Wolf S, Beachy RN, Lucas WJ (1992) Secondary plasmodesmata are specific sites of localization of the tobacco mosaic virus movement protein in transgenic tobacco plants. Plant Cell 4: 915–928
— —, Willmitzer L, Lucas WJ (1993) Correlation between arrested secondary plasmodesmal development and onset of accelerated leaf senescence in yeast acid invertase transgenic tobacco plants. Plant J 4: 179–189
Ehlers K, Kollmann R (1996) Formation of branched plasmodesmata in regeneratingSolatium nigrum protoplasts. Planta 199: 126–138
Heslop-Harrison J (1966) Cytoplasmic continuities during spore formation in flowering plants. Endeavour 25: 65–72
Jamieson JD, Palade G (1967) Intracellular transport of secretory protein in the pancreatic exocrine cells. J Cell Biol 34: 577
Jones MGK (1976) The origin and development of plasmodesmata. In: Gunning BES, Robards AW (eds) Intercellular communication in plants: studies on plasmodesmata. Springer, Berlin Heidelberg New York, pp 81–105
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixation of high osmolality for use in electron microscopy. J Cell Biol 27: 137A
Karp G (1996) Cell and molecular biology. Wiley, New York
Kollmann R, Glockmann C (1985) Studies on graft unions I: plasmodesmata between cells of plants belonging to different unrelated taxa. Protoplasma 124: 224–235
Kollmann R, Glockmann C (1991) Studies on graft unions III: on the mechanism of secondary formation of plasmodesmata at the graft interface. Protoplasma 165: 71–85
—, Yang S, Glockmann C (1985) Studies on graft unions II: continuous and half plasmodesmata in different regions of the graft interface. Protoplasma 126: 19–29
Lucas WJ, Ding B, van der School C (1993) Plasmodesmata and the supracellular nature of plants. New Phytol 125: 435–476
Mezitt LA, Lucas WJ (1996) Plasmodesmal cell-to-cell transport of proteins and nucleic acids. Plant Mol Biol 32: 251–273
Nessler CL, Mahlberg PG (1981) Cytochemical localization of cellulase activity in articulated, anastomosing laticifers ofPapaver somniferum L. (Papaveraceae). Am J Bot 68: 730–732
Nie SW, Wang XY, Cheng KC (1984) Transmission and scanning electron microscopic observation on the plasmodesmal channels between the pollen mother cells of lily. Acta Bot Sin 26: 34–37
Robards AW, Lucas WJ (1990) Plasmodesmata. Annu Rev Plant Physiol Plant Mol Biol 41: 369–419
Wu, BJ, Xie, MT, Cui, YY, Chen, YP, Guo, WH, Jiang, H, Cheng, KC (1995) Formation of secondary plasmodesmata and intercellular passages by means of ACHT. Sci China Ser B 38: 187–194
Zheng GC (KC Cheng), Yang CL, Zheng YR (1987) The relationship between cytomixis, chromosome mutation and karyotype evolution on lily. Caryologia 40: 243–259
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X.Y., Guo, G.Q., Nie, X.W. et al. Cytochemical localization of cellulase activity in pollen mother cells of david lily during meiotic prophase I and its relation to secondary formation of plasmodesmata. Protoplasma 204, 128–138 (1998). https://doi.org/10.1007/BF01280319
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01280319