Summary
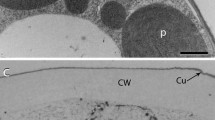
The ultrastructure of developing cotton fibers has been examined using novel modifications of the techniques of surface replication, freeze-etching and thin-sectioning. The fiber surface was found to be coated with a lamellar cuticle, which is stretched and thinned as the fiber elongates. It is marked by bars which run parallel with the fiber long axis. Beneath the cuticle, the outermost microfibrils of the primary wall lie parallel with the fiber axis, while those adjacent to the plasma membrane are transverse. Primary wall microfibrils are present in bundles, disposed in left-handed and right-handed helices, which correspond with the fibrils observed optically. Microfibrils within bundles form in-phase waves, with wavelengths and amplitudes in the ranges 0.3–7 μm and 0.01–0.1 μm in primary and secondary walls respectively. As elongation proceeds bundles become displaced towards the cell axis. Microfibrils of the secondary wall, disposed around the cell as fast helices, are similarly bundled and wavy (though with a reduced amplitude). In surface-replicas, large (20–30 nm) granules are present on the cytoplasmic face of the wall which probably correspond with 20–40 nm low prominences visible on freeze-etch EF plasma membrane fracture faces. It is proposed that these may be microfibril-synthesizing centers. Plasma membranes fracture such that the membrane-associated-particles segregate 60∶40 between P and E fracture moieties, but the prominence and total number of these particles is reduced at the stage of secondary wall formation as compared with primary wall formation. Beneath the plasmalemma the axes of microtubules parallel secondary wall microfibril orientation. Cross-bridges, which stain heavily after glutaraldehyde/tannic acid fixation, link microtubules to plasma membrane. The use of butyl benzene to cement fragments of cotton fibers, employed in this work, may prove useful in other freeze-etch studies of long fibers which are readily ruptured during preparation.
Similar content being viewed by others
References
Aiyangar, G. S., 1951: Origin and development of lint and fuzz in cotton. Indian J. Agr. Sci.21, 293–312.
Anderson, D. B., andT. Kerr, 1938: Growth and structure of cotton fibre. Ind. Eng. Chem.30, 48–54.
Bachmann, L., andW. W. Schmitt, 1971: Improved cryofixation applicable freeze-etching. Proc. nat. Acad. Sci. (U.S.A.)68, 2149–2152.
Balls, W. L., 1923: The determiners of cellulose structure as seen in the cell-wall of cotton hairs. Proc. roy. Soc. (Lond.)B 95, 72–89.
—, andH. A. Hancock, 1922: Further observations on cell-wall structure as seen in cotton hairs. Proc. roy. Soc. (Lond.)B 93, 426–440.
Boyd, J. D., andR. C. Foster, 1975: Microfibrils in primary and secondary wall growth develop trellis configurations. Can. J. Bot.53, 2687–2701.
Branton, D., S. Bullivant, N. B. Gilula, M. Karnovsky, H. Moor, K. Mühlethaler, D. H. Northcote, L. Packer, B. Satir, P. Satir, V. Speth, L. A. Staehelin, R. L. Steere, andR. S. Weinstein, 1975: Freeze-etching nomenclature. Science190, 54–56.
—, andD. W. Deamer, 1972: Membrane structure. In: Protoplasmatologia II/E/1. Wien-New York: Springer.
Brown, R. M., Jr., andD. Montezinos, 1976: Cellulose microfibrils: visualization of biosynthetic and orienting complexes in association with the plasma membrane. Proc. nat. Acad. Sci. (U.S.A.)73, 143–147.
Hanke, D. E., andD. H. Northcote, 1974: Cell wall formation by soybean callus protoplasts. J. Cell Sci.14, 29–50.
Heath, I. B., 1974: A unified hypothesis for the role of membrane bound enzyme complexes and microtubules in plant cell wall synthesis. J. theor. Biol.48, 445–449.
Houwink, A. C., andP. A. Roelofsen, 1954: Fibrillar architecture of growing cell walls. Acta bot. Neerl.3, 385–395.
Itoh, T., 1974: Fine Structure and formation of cell wall of developing cotton fiber. Wood Res.56, 49–61.
Kinsinger, W. G., andC. W. Hock, 1948: Electron microscopical studies of natural cellulose fibers. Ind. Eng. Chem.40, 1711–1716.
Marx-Figini, M., andG. V. Schulz, 1966: Über die Kinetik und den Mechanismus der Biosynthese der Cellulose in den Höheren Pflanzen (nach Versuchen an den Samenhaaren der Baumwolle). Biochim. biophys. Acta112, 81–101.
Meinert, M., andD. Delmer, 1976: Biochemical changes in cell wall composition associated within vivo andin vitro fiber development inGossypium hirsutum. Pl. Physiol.57, Suppl. 291 A.
Mizuhira, V., andY. Futaesaku, 1972: New fixation for biological membranes using tannic acid. Acta Histochem. Cytochem.5, 233–235.
Montezinos, D., and R. M.Brown, Jr., 1977: Surface architecture of the plant cell: biogenesis of the cell wall, with special emphasis on the role of the plasma membrane in cellulose biosynthesis. J. supramolec. Struct.: in press.
Moor, H., andK. Mühlethaler, 1963: Fine structure in frozen etched yeast cells. J. Cell Biol.17, 609–628.
Mueller, S., R. M. Brown Jr., andT. K. Scott, 1976: Cellulosic microfibrils: nascent stages of synthesis in a higher plant cell. Science194, 949–951.
Mühlethaler, K., 1949: Electron micrographs of plant fibers. Biochim. biophys. Acta3, 15–25.
Newcomb, E. H., 1969: Plant microtubules. Ann. Rev. Plant Physiol.20, 253–288.
Northcote, D. H., 1972: Chemistry of the plant cell wall. Ann. Rev. Plant Physiol.23, 113–132.
O'Kelley, J. C., 1953: The use of14C in locating growth regions of cell walls of elongating cotton fibers. Plant Physiol.28, 281–286.
—, andP. H. Carr, 1953: Elongation of the cotton fiber. In: Growth and differentiation in plants (Loomis, W., ed.), pp. 55–68. Ames, Iowa: Iowa State College Press.
Preston, R. D., 1959: Wall organization in plant cells. Int. Rev. Cytol.8, 33–60.
—, 1964: Structural and mechanical aspects of plant cell walls with particular reference to synthesis and growth. In: Formation of wood in forest trees (Zimmermann, M., ed.), pp. 168–188. New York: Academic Press.
Ramsey, J. D., andJ. D. Berlin, 1976: Ultrastructure of early stages of cotton fiber differentiation. Bot. Gaz.137, 11–19.
Rånby, B. G., undE. Ribi, 1950: Über den Feinbau der Zellulose. Experientia6, 12–14.
Roelofsen, P. A., 1965: Ultrastructure of the wall in growing cells and its relation to the direction of growth. Advan. bot. Res.2, 69–149.
—, 1958: Cell-wall structure as related to surface growth. Acta bot. Neerl.7, 77–89.
—, andA. L. Houwink, 1953: Architecture and growth of primary cell wall in some plant hairs and in thePhycomyces sporangiophore. Acta bot. Neerl.2, 218–225.
Roland, J. C., B. Vian, andD. Reis, 1975: Observations with cytochemistry and ultracryotomy on the fine structure of the expanding walls in actively elongating plant cells. J. Cell Sci.19, 239–259.
Rollins, M. L., 1968: Cell wall structure and cellulose synthesis. For. Prod. J.18 (2), 91–100.
Schubert, A. M., C. R. Benedict, J. D. Berlin, andR. J. Kohel, 1973: Cotton fiber development — Kinetics of cell elongation and secondary wall thickening. Crop Sci.13, 704–709.
Waterkyn, L., E. DeLanghe, andA. A. H. Eid, 1975:In vitro culture of fertilized cotton ovules. La Cellule,71, 39–52.
Westafer, J. M., andR. M. Brown, Jr., 1976: Electron microscopy of the cotton fiber: new observations on cell wall formation. Cytobios15, 111–138.
Willison, J. H. M., 1975: Plant cell-wall microfibril disposition revealed by freeze-fractured plasmalemma not treated with glycerol. Planta (Berl.)126, 93–96.
—, 1976: An examination of the relationship between freeze-fractured plasmalemma and cell-wall microfibrils. Protoplasma88, 187–200.
- and R. M.Brown, Jr., 1977: In preparation.
—, andE. C. Cocking, 1975: Microfibril synthesis at the surfaces of isolated tobacco mesophyll protoplasts. Protoplasma84, 147–159.
—, andB. W. W. Grout, 1973: Colloq. Int. C.N.R.S.212, 313.
Wuhrmann, K., A. Heuberger, andK. Mühlethaler, 1946: Elektronenmikroskopische Untersuchungen an Zellulosefasern nach Behandlung mit Ultraschall. Experientia6, 12–14.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Willison, J.H.M., Brown, R.M. An examination of the developing cotton fiber: Wall and plasmalemma. Protoplasma 92, 21–41 (1977). https://doi.org/10.1007/BF01280198
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01280198