Summary
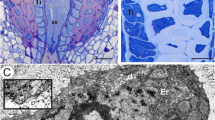
Symplasmic contacts of Strasburger cells in the mature needle ofMetasequoia glyptostroboides were analysed with special regard to changes of plasmodesmata in fine structure and distribution. In meristematic cells simple primary plasmodesmata are evenly distributed throughout the entire wall, whereas in mature Strasburger cells plasmodesmata are aggregated in defined, dome-shaped wall thickenings. The elongated, often multiple-branched cytoplasmic strands show a distinct neck region besides a considerably dilated sleeve region confluent with cavities, which have formed at branching sites of plasmodesmata in various planes of the wall thickening. Most branches radiating from these cavities connect the protoplasts of the adjacent cells; occasionally some strands are discontinuous. The desmotubules of both, continuous and discontinuous plasmodesmal branches exhibit great variability in structure and number: they may be partially dilated, multiple-stranded and branched within single plasmodesmal branches. Fine structurally, plasmodesmata of Strasburger cells show great resemblance with developing sieve pores of conifers. This characteristic fine structure implicates a special role of the endomembrane system for phloem loading in theMetasequoia leaf.
Similar content being viewed by others
References
Barnett JR (1982) Plasmodesmata and pit development in secondary xylem elements. Planta 155: 251–260
— (1987a) The development of fibre-tracheid pit membranes inPyrus communis L. IAWA Bull ns 8: 134–142
— (1987b) Changes in the distribution of plasmodesmata in developing fibre-tracheid pit membranes ofSorbus aucuparia L. Ann Bot 59: 269–279
Behnke H-D, Paliwal GS (1973) Ultrastructure of phloem and its development inGnetum gnemon, with some observations onEphedra campylopoda. Protoplasma 78: 305–319
Blechschmidt-Schneider S, Einig W, Hampp R (1995) Symplastic loading in spruce needles. In: International Conferences on the Transport of Photoassimilates, Canterbury, Kent, UK, 1995, abstract no 3.13
Campbell R (1972) Electron microscopy of the development of needles ofPinus nigra var.maritima. Ann Bot 36: 711–720
Carde J-P (1974) Le tissu de transfert (= cellules de Strasburger) dans les aiguilles du pin maritime (Pinus pinaster Ait.). II. Caractères cytochimiques et infrastructuraux de la paroi et des plasmodesmes. J Microsc 20: 51–72
Currier HB (1957) Callose substances in plant cells. Am J Bot 44: 478–488
Ding B, Lucas WJ (1996) Secondary plasmodesmata: biogenesis, special functions, and evolution. In: Smallwood M, Knox JP, Bowles DJ (eds) Membranes: specialized functions in plants. Bios Scientific Publishers, Oxford, pp 489–506
Ehlers K, Kollmann R (1996) Formation of branched plasmodesmata in regeneratingSolanwn nigrum protoplasts. Planta 199: 126–138
Evert RF, Bornman ChH, Butler V, Gilliland MG (1973) Structure and development of sieve areas in leaf veins ofWelwitschia. Protoplasma 76: 23–34
Gamalei YV (ed) (1990) Leaf phloem. Nauka, Leningrad (in Russian)
—, van Bel AIE, Pakhomova MV, Sjutkina AV (1994) Effects of temperature on the conformation of the endoplasmic reticulum and on starch accumulation in leaves with the symplasmic minor-vein configuration. Planta 194: 443–453
Gambles RL, Dengler RE (1982) The anatomy of the leaf of red pine,Pinus resinosa. II. Vascular tissues. Can J Bot 60: 2804–2824
Grabski S, de Feijter AW, Schindler M (1993) Endoplasmic reticulum forms a dynamic continuum for lipid diffusion between contiguous soybean root cells. Plant Cell 5: 25–38
Hughes J, McCully ME (1975) The use of an optical brightener in the study of plant structure. Stain Technol 50: 319–329
Jones MGK (1976) The origin and development of plasmodesmata. In: Gunning BES, Robards AW (eds) Intercellular communication in plants: studies on plasmodesmata. Springer, Berlin Heidelberg New York, pp 81–103
Kollmann R (1973) Cytologie des Phloems. In: Hirsch GC, Ruska H, Sitte P (eds) Grundlagen der Zytologie. G Fischer, Jena, pp 479–505
—, Glockmann C (1991) Studies on graft unions. III. On the mechanism of secondary formation of plasmodesmata at the graft interface. Protoplasma 165: 71–85
—, Schumacher W (1963) Über die Feinstruktur des Phloems vonMetasequoia glyptostroboides und seine jahreszeitlichen Veränderungen. IV. Mitteilung. Weitere Beobachtungen zum Feinbau der Plasmabrücken in den Siebzellen. Planta 60: 360–389
— — (1964) Über die Feinstruktur des Phloems vonMetasequoia glyptostroboides und seine jahreszeitlichen Veränderungen. V. Mitteilung. Die Differenzierung der Siebzellen im Verlaufe einer Vegetationsperiode. Planta 63: 155–190
Lane BP, Europa DL (1965) Differential staining of ultrathin sections of Epon-embedded tissues for light microscopy. J Histochem Cytochern 13: 579–582
Lucas WJ, Gilbertson RL (1994) Plasmodesmata in relation to viral movement within leaf tissues. Annu Rev Phytopathol 32: 387–411
Neuberger DS, Evert RF (1975) Structure and development of sieve areas in the hypocotyl ofPinus resinosa. Protoplasma 84: 109–125
Offler CE, Patrick JW (1992) The role of symplastic transport of assimilates in developing reproductive sinks. In: Basic and applied research in plasmodesmatal biology. 2nd International Workshop, Oosterbeek, The Netherlands, 1992, pp 105–108
Parameswaran N, Liese W (1970) Zur Cytologie der Strasburger-Zellen in Coniferennadeln. Naturwissenschaften 57: 45–46
— — (1981) Torus-like structures in interfibre pits ofPrunus andPyrus. IAWA Bull ns 2: 89–93
Russin WA, Evert RF (1985) Studies on the leaf ofPopulus deltoides (Salicaceae): ultrastructure, plasmodesmatal frequency, and solute concentrations. Am J Bot 72: 1232–1247
Sauter JJ, Dörr I, Kollmann R (1976) The ultrastructure of Strasburger cells (= albuminous cells) in the secondary phloem ofPinus nigra var.austriaca (Hoess) Badoux. Protoplasma 88: 31–49
Schindler M (1992) Symplastic views of a complex organelle. Meeting Report. Plant Cell 4: 1467–1469
Schmilz K, Srivastava LM (1974) Fine structure and development of sieve tubes inLaminaria groenlandica Rosenv. Cytobiologie 10: 66–87
Schulz A (1990) Conifers. In: Behnke H-D, Sjolund RD (eds) Sieve elements: comparative structure, induction and development. Springer, Berlin Heidelberg New York Tokyo, pp 63–86
— (1992) Living sieve cells of conifers as visualized by confocal, laser-scanning fluorescence microscopy. Protoplasma 166: 153–164
Singh AP (1984)Pinus radiata needle trace studies: fine structure of immature sieve cells in the primary phloem. Cytologia 49: 359–384
Steinberg G, Kollmann R (1994) A quantitative analysis of the interspecific plasmodesmata in the non-division walls of the plant chimeraLaburnocytims adamii (Poit.) Schneid. Planta 192: 75–83
Strasburger E (1891) Über den Bau und die Verrichtungen der Leitungsbahnen in den Pflanzen. In: Strasburger E (ed) Leitungsbahnen in den Pflanzen. G Fischer, Jena, p 55
Warmbrodt RD, Eschrich W (1985) Studies on the mycorrhizas ofPinus sylvestris L. produced in vitro with the basidiomyceteSuillus variegatus (Sw. ex Fr.) O. Kuntze. II. Ultrastructural aspects of the endodermis and vascular cylinder of the mycorrhizal rootlets. New Phytol 100: 403–418
Wooding FBP (1968) Fine structure of callus phloem inPinus pinea. Planta 83: 99–110
— (1974) Development and fine structure of angiosperm and gymnosperm sieve tubes. In: Sleigh MA, Jennings DH (eds) Transport at the cellular level. Cambridge University Press, Cambridge, pp 27–41 (Society of Experimental Biology symposia, vol 28)
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. Dr. h.c. Eberhard Schnepf on the occasion of his retirement
Rights and permissions
About this article
Cite this article
Glockmann, C., Kollmann, R. Structure and development of cell connections in the phloem ofMetasequoia glyptostroboides needles I. Ultrastructural aspects of modified primary plasmodesmata in Strasburger cells. Protoplasma 193, 191–203 (1996). https://doi.org/10.1007/BF01276645
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01276645