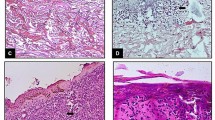
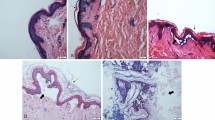
Abstract
The temporal course of epidermal basal cell proliferation in the wound of the pinna of rats was studied using bromodeoxyuridine (BrdU) immunohistochemistry. Following incisional wounding, the animals were sacrificed at intervals ranging from 0 hours to 32 days. Two biopsies were taken from each animal, one intravitally and one postmortem after 24 hours storage at 8°C. Specimens were incubated in a solution containing BrdU and embedded in paraffin. BrdU expression was demonstrated by a monoclonal antibody against BrdU. In both intravital and postmortem biopsies, the labelling indices increased significantly in the period from 32 to 60 hours post-injury. This suggests that DNS synthesis induced during life continues after death. Applied to forensic practice, the present findings point to the possibility of determining the vitality of a wound in postmortem tissue.
Zusammenfassung
Im Tierexperiment wurde die Zeitabhängigkeit der Proliferationsaktivität von epidermalen Basalzellen aus der Schnittwunde des Rattenohres unter Verwendung von BrdU (in vitro-Inkubation) durch Bestimmung des Markierungsindexes untersucht. Nach Schnittverletzung überlebten die Tiere unterschiedliche Zeitintervalle von 0 Stunden bis 32 Tage. Es erfolgten pro Tier je 2 Biopsieentnahmen: vital und 24 Stunden nach Eintritt des Todes (postmortal), währenddessen der Tierkörper bei 8°C im Kühlschrank gelagert wurde. Folgendes Ergebnis konnte gewonnen werden: Unter der Annahme eine p ≥ 0.01 nahm der Markierungsindex epidermaler Basalzellen nach 32 Stunden Überlebenszeiten bis zur 60. Stunde nach Wundsetzung signifikant zu. Diese Zunahme war nach vitaler Biopsie ebenso wie nach postmortaler Biopsie nachweisbar. Es ist somit davon auszugehen, daß nach der einmal erfolgten Proliferationsinduktion sich diese auch postmortal fortsetzt. Für die forensische Praxis ist festzustellen, daß auch an postmortalem Gewebe mindestens eine Vitalitätsbestimmung vorgenommen werden dürfte.
Similar content being viewed by others
References
Betz P, Nerlich A, Wilske J, Tübel J, Penning R, Eisenmenger W (1993) The time-dependent localization of Ki67 antigen-positive cells in human skin wounds. Int J Leg Med 106:35–40
Brown DC, Garter KC (1990) Monoclonal antibody Ki67: its use in histopathology. Histopathology 17:489–503
Bullough WS (1973) The epidermal chalone. Natl Cancer Inst Monogr 38:94–107
Bullough WS, Laurence EB (1960) The control of mitotic activity in mouse skin. Exp Cell Res 21:394–405
Christophers E, Braun-Falco O (1967) Epidermale Regeneration am Meerschweinchenohr nach Hornhautabriß. Eine autoradiographische Untersuchung. Arch Klin Exp Dermatol 231:85–96
Frasunek J, Oehmichen M (1986) Postmortale Cytokinetic epidermaler Zellen. Beitr Gerichtl Med 44:321–324
Gerdes J, Li L, Schlueter C, Duchrow M, Wohlenberg C, Gerlach C, Stahmer I, Kloth S, Brandt E, Flad H-D (1991) Immunobiochemical and molecular biologic characterization of the cell proliferation-associated nuclear antigen that is defined by monoclonal antibody Ki-67. Am J Pathol 138:867–873
Gerdes J, Becker M, Key G, Cattoretti G (1992) Immunohistologic detection of tumor growth factor (Ki67-antigen) in formalin fixed and routinely processed tissue. J Pathol 168:85–87
Gratzner HG (1982) Monoclonal antibody to 5-bromo- and 5-iododeoxyuridine: a new reagent for detection of DNA replication. Science 218:474–475
Hardonk MJ, Harms G (1990) The use of 5′-bromodeoxyuridine in the study of cell proliferation. Acta Histochem [Suppl] 39: 99–108
Hell EA, Cruickshank CND (1963) The effect of injury upon uptake of3H-thymidine by guinea pig epidermis. Exp Cell Res 31:128–139
Hume WJ (1990) A reproducible technique combining tritiated thymidine autoradiography with immunodetection of bromodeoxyuridine for double labelling studies of cell proliferation in paraffin sections of tissues. Cell Tissue Kinet 23:161–168
Iversen OH (1981) The chalones. In: Baserga R (ed) Tissue growth factors Springer, Berlin Heidelberg New York, pp 491–550
Jones HB, Clarke NAB, Barrass NG (1993) Phenobarbital-induced hepatocellular proliferation: anti-bromodeooxyuridine and antiproliferating cell nuclear antigen immunocytochemistry. J Histochem Cytochem 41:21–27
Lin P, Allison DC (1993) Measurement of DNA content and tritiated thymidine and bromodeoxyuridine incorporation by the same celle. J Histochem Cytochem 41:1435–1439
McDowell EM, Zhang X-M, DeSanti AM (1990) Localization of healing tracheal wounds using bromodeoxyuridine immunohistochemistry. Stain Technol 65:25–29
Nowakowski RS, Lewin SB, Miller MW (1989) Bromodeoxyuridine immunohistochemical determination of the lengths of the cell cycle and the DNA-synthetic phase for an anatomically defined population. J Neurocytol 18:311–318
Odland G, Ross R (1968) Human wound repair. I. Epidermal regeneration. J Cell Biol 39:135–151
Oehmichen M (1990) Die Wundheilung. Springer, Berlin Heidelberg New York
Oehmichen M, Frasunek J, Zilles K (1988a) Cytokinetics of epidermal cells in skin from human cadavers: I. Dependency on the postmortal interval. Z Rechtsmed 101:161–171
Oehmichen M, Frasunek J, Zilles K (1988b) Cytokinetics of epidermal cells in skin from human cadavers: II. Dependency on sex, age and site. Z Rechtsmed 101:173–182
Oehmichen M, Zilles K (1984) Postmortale DNS- und RNS-Synthese: Erste Untersuchungen an menschlichen Leichen. Z Rechtsmed 91:287–294
Oostrum IEA van, Rozemuller E, Knol RFG, Erkens-Schulze S (1990) Cell proliferation kinetics of six xenografted human cervix carcinomas: comparison of autoradiography and bromodeoxyuridine labelling methods. Cell Tissue Kinet 23:523–544
Parkins CS, Bush C, Price P, Steel GG (1991) Cell proliferation in human tumour xenografts: measurement using antibody labelling against bromodeoxyuridine and Ki-67. Cell Prolif 24:171–179
Repka AM, Adler R (1992) Accurate determination of the time of cell birth using a sequential labelling technique with3H-thymidine and bromodeoxyuridine (“window labeling”). J Histochem Cytochem 40:947–953
Roels H (1981) Hyperplasia versus atrophy — regeneration versus repair. In: Glynn LE (ed) Tissue and regeneration. Elsevier, Amsterdam, pp 243–283
Sachs L (1984) Angewandte Statistik, 6. Auf, Springer, Berlin Heidelberg New York, Tokyo
Schellmann B (1981) Postmortaler Einbau von3H-Thymidin in die Epidermis und Mundschleimhaut des Menschen. Habilitationsschrift. Universität Erlangen
Schultze B (1968) Die Orthologie und Pathologie des Nucleinsäure- und Eiweißstoffwechsels der Zelle im Autoradiogramm. In: Handbuch der allgemeinen Pathologie, H.W. Altmann et al. (eds.), Vol 2, part 5 (Die Zelle), pp. 466–670. Springer, Berlin Heidelberg New York
Van der Laan M, Thomas CB (1985) Characterization of monoclonal antibodies to bromodeoxyuridine. Cytrometry 6:501–505
Wheeler J, Matthews JNS, Morley AR (1991) Corrections in counting paired nuclei labelled with bromodeoxyuridine in tissue sections. Cell Prolif 24:143–157
White RA, Terry NHA, Meistrich ML (1990) New methods for calculating kinetic properties of cells in vitro using pulse labelling with bromodeoxyuridine. Cell Tissue Kinet 23:561–573
Zeymer U, Fishbein MC, Forrester IS, Cercek B (1992) Proliferating cell nuclear antigen immunohitochemistry in rat aorta after balloon denudation. Comparison with thymidine and bromodeoxyuridine labelling. Am J Pathol 141:685–690
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oehmichen, M., Cröpelin, A. Temporal course of intravital and postmortem proliferation of epidermal cells after mechanical injury. Int J Leg Med 107, 257–262 (1995). https://doi.org/10.1007/BF01245484
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01245484