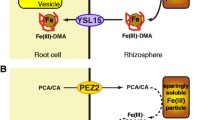
Summary
We have investigated the mugineicacid-Fe transport activity of Fe-deficient barley roots, using the multi-compartment transport box system. The roots maintained Fe transport activity for 20 h after excision. The following results were obtained. (1) In Fe-deficient roots, mugineic acid addition enhanced the transport of Fe by 32.2 times over that of the control (with FeC13 addition). (2) The mugineic-acid-55Fe transport activity of Fe-deficient roots was 18.4-fold higher than that of the Fe-sufficient roots. (3) The mugineic-acid-55Fe transport activity was decreased (7.13% based on the control) by treatment with 5 μM carbonylcyanidem-chlorophenyl hydrazone (CCCP). Pretreatment with 0.1 mM dicyclohexyl carbodiimide (DCCD) lowered the transport activity (10.7% based on the control) and 1 mMN-ethylmaleimide (NEM) pretreatment reduced the transport activity to a value equivalent to 2.41% of that in the control. It is concluded that mugineicacid-Fe transporter is induced in its activity and/or amount by Fe-deficiency treatment and has an SH residue at its active site, and that the transporter needs the proton motive force produced by ATPase. We detected three polypeptides (14, 28 and 40 kDa) in the root plasma membrane that were induced under Fe-deficiency treatment.
Similar content being viewed by others
Abbreviations
- p-APMSF:
-
(p-amidinophenyl)methanesulfonyl fluoride hydrochloride
- CCCP:
-
carbonylcyanide m-chlorophenylhydrazone
- DCCD:
-
dicyclohexylcabodiimide
- DMSO:
-
dimethyl sulfoxide
- MA:
-
mugineic acid
- NEM:
-
N-ethylmaleimide
References
Clarkson DT, Sanderson J (1978) Sites of absorption and translocation of iron in barley roots. Plant Physiol 61:731–736
Hurkman WJ, Tanaka CK (1986) Solubilization of plant membrane proteins for analysis by two-dimensional gel electrophoresis. Plant Physiol 81:802–806
Jolley VD, Brown JC, Blaylock MJ, Camp SD (1988) A role for potassium in the use of iron by plants. J Plant Nutr 11:1159–1175
Kawasaki T, Shimizu G, Moritsugu M (1983) Effects of high concentrations of sodium chloride and polyethylene glycol on the growth and ion absorption in plants, II. Multi-compartment transport box experiment with excised roots of barley. Plant Soil 75:87–93
Larsson C (1985) Plasma membranes Mod Methods Plant Anal New Ser 1:85–104
Marschner H, Römheld V, Kissel M (1987) Localization of phytosiderophore release and of iron uptake along intact barley roots. Physiol Plant 71:157–162
Mihashi S, Mori S, Nishizawa N (1989) Enhancement of mugineic-acid-Fe3+ uptake by Fe-deficient barley roots with coexistence of excess free mugineic acid in the medium. Plant Soil, in press
Mori S, Nishizawa N (1987) Methionine as a dominant precursor of phytosiderophores in Graminaceae plants. Plant Cell Physiol 28:1081–1092
Mori S, Nishizawa N, Kawai S, Sato Y, Takagi S (1987) Dynamic state of mugineic acid and analogous phytosiderophores in Fe-deficient barley. J Plant Nutr 10:1003–1011
Nagahashi G, Leonard RT, Thomson WW (1978) Purification of plasma membranes from roots of barley. Plant Physiol 61:993–999
Reinhold L, Kaplan A (1984) Membrane transport of sugars and amino acids. Annu Rev Plant Physiol 35:45–83
Takagi S (1984) Fe uptake regulation system in root and its differences among plant species. In: Japanese society of soil science and plant nutrition (ed). Recent problems and studies on soil science and fertilizer in Tohoku region. pp 190–195 (in Japanese)
Takagi S, Nomoto K, Takemoto T (1984) Physiological aspect of mugineic acid, a possible phytosiderophore of graminaceous plants. J Plant Nutr 7:469–477
Yoshida S, Uemura M, Niki T, Sakai A, Gusta LV (1983) Partition of membrane particles in aqueous two-polymer phase system and its practical use for purification of plasma membranes from plants. Plant Physiol 72:105–114
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mihashi, S., Mori, S. Characterization of mugineic-acid-Fe transporter in Fe-deficient barley roots using the multi-compartment transport box method. Biol Metals 2, 146–154 (1989). https://doi.org/10.1007/BF01142553
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01142553