Abstract
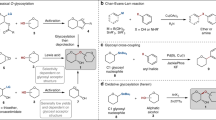
Three chemical transformations of oligosaccharide 1-deoxy-1-(4-trifluoroacetamidophenyl)aminoalditols are described. 1) Oxidation with hydrogen peroxide to give the corresponding reducing oligosaccharides. 2) Oxidation with cerium ammonium nitrate to give the corresponding 1-amino-1-deoxyalditols. 3) Treatment with acetic anhydride to give the correspondingN-acetylated derivatives, which are more stable towards oxidation.
Similar content being viewed by others
References
Kallin E, Lönn H, Norberg T (1986) Glycoconjugate J 3:311–19.
Wiegandt H, Ziegler W (1974) Hoppe-Seylers Z Physiol Chem 355:11–18.
Baumgarten RJ (1966) J Chem Ed 43:398–408 and references cited therein.
Hoffman RV, Kumar A (1984) J Org Chem 49:4011–14.
De La Mare H (1960) J Org Chem 25:2114–26.
Hünig S, Daum W (1955) Justus Liebigs Ann Chem 595:131–55.
Mason LFA (1975) in Photographic Processing Chemistry, Focal Press, London, p 225–55.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kallin, E., Lönn, H. & Norberg, T. Derivatization procedures for reducing oligosaccharides, part 2: Chemical transformation of 1-deoxy-1-(4-trifluoroacetamidophenyl)aminoalditols. Glycoconjugate J 5, 145–150 (1988). https://doi.org/10.1007/BF01061203
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01061203