Abstract
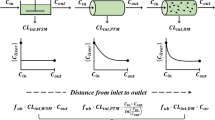
The potential influence of protein binding kinetics on elimination from liver sinusoids was evaluated by means of a “well-stirred” model (I) and a “tube” model (II). When the dissociation rate constant (k−1) is at the estimated maximum, equilibrium is maintained during the passage of drug through the eliminating organ, and hence dissociation as such has no limiting effect on elimination. When, however, k−1 is at the estimated minimum, equilibrium is not maintained, the unbound fraction is reduced during the passage, and a significant decrease in the extraction ratio occurs when the unbound fraction is 0.01 or less. The models were furthermore used to investigate the effect of saturation, of both the binding protein and the elimination process, on elimination.
Similar content being viewed by others
References
E. Kr:uger-Thiemer, W. Diller, and P. B:unger. Pharmacokinetic models regarding protein binding of drugs.Antimicrob. Agents Chemother. 1965:183–191 (1966).
B. K. Martin. Kinetics of elimination of drugs possessing high affinity for the plasma proteins.Nature 207:959–960 (1965).
J. J. Coffey, F. J. Bullock, and P. T. Schoenemann. Numerical solution of nonlinear pharmacokinetic equations: effects of plasma protein binding on drug distribution and elimination.J. Pharm. Sci. 60:1623–1628 (1971).
J. G. Wagner. Simple model to explain effects of plasma protein binding and tissue binding on calculated volumes of distribution, apparent elimination rate constants and clearances.Eur. J. Clin. Pharmacol. 10:425–432 (1976).
P. J. McNamara, G. Levy, and M. Gibaldi. Effect of plasma protein and tissue binding on the time course of drug concentration in plasma.J. Pharmacokin. Biopharm. 7:195–206 (1979).
K. B. Bischoff and R. L. Dedrick. Thiopental pharmacokinetics.J. Pharm. Sci. 57:1346–1351 (1968).
M. Rowland, L. Z. Benet, and G. G. Graham. Clearance concepts in pharmacokinetics.J. Pharmacokin. Biopharm. 1:123–135 (1973).
G. R. Wilkinson and D. G. Shand. A physiological approach to hepatic drug clearance.Clin. Pharmacol. Ther. 18:377–390 (1975).
K. S. Pang and M. Rowland. Hepatic clearance of drugs. I. Theoretical considerations of a “well-stirred” model and a “parallel tube” model. Influence of hepatic blood flow, plasma, and blood cell binding, and the hepatocellular enzymatic activity on hepatic drug clearance.J. Pharmacokin. Biopharm. 5:625–653 (1977).
K. Winkler, L. Bass, S. Keiding, and N. Tygstrup. The effect of hepatic perfusion on assessment of kinetic constants. In F. Lundquist and N. Tygstrup (eds.),Alfred Benzon Symposium VI: Regulation of Hepatic Metabolism, Munksgaard, Copenhagen, 1974, pp. 797–807.
L Bass, S. Keiding, K. Winkler, and N. Tygstrup. Enzymatic elimination of substrates flowing through the intact liver.J. Theor. Biol. 61:393–409 (1976).
H. Gutfreund.Enzymes: Physical Principles. Wiley-Interscience, London, 1972, p. 158.
C. A. Goresky. A linear method for determining liver sinusoidal and extravascular volumes.Am. J. Physiol. 204:626–640 (1963).
R. W. Flower and B. F. Hochheimer. Clinical technique and apparatus for simultaneous angiography of retinal and choroidal circulations.Invest. Opththalmol. 12:248–261 (1973).
G. R. Cherrick, S. W. Stein, C. M. Leevy, and C. S. Davidson. Indocyanine green: observations on its physical properties, plasma decay, and hepatic extraction.J. Clin. Invest. 39:592–600 (1960).
J. Caesar, S. Shaldon, L. Chiandussi, L. Guevara, and S. Sherlock. The use of indocyanine green in the measurement of hepatic blood flow and as a test of hepatic function.Clin. Sci. 21:43–57 (1961).
C. M. Leevy, C. L. Mendenhall, W. Lesko, and M. M. Howard. Estimation of hepatic blood flow with indocyanine green.J. Clin. Invest. 41:1169–1179 (1962).
L. Vettore, G. Falezza, M. C. de Matteis, G. Cetto, and M. Zandegiacomo. A new method for the determination of sodium and potassium in human red blood cells, using indocyanine green as a marker for trapped plasma.Clin. Chim. Acta 55:345–351 (1974).
K. J. Baker. Binding of sulfobromophthalein (BSP) sodium and indocyanine green (ICG) by plasmaα 1-lipoproteins.Proc. Soc. Exp. Biol. Med. 122:957–963 (1966).
S. D. Stroupe and U. Westphal. Steroid-protein interactions. Stopped-flow fluorescence studies of the interaction between steroid hormones and progesterone-binding globulin.J. Biol. Chem. 250:8735–8739 (1975).
F. B. Freedman and J. A. Johnson. Equilibrium and kinetic properties of the Evans blue-albumin system.Am. J. Physiol. 216:675–681 (1969).
A. Froese and A. H. Sehon. Kinetic studies of protein-dye and antibody-hapten interactions with the temperature-Jump method.Can. J. Chem. 40:1786–1797 (1962).
J. A. Jansen. Kinetics of the binding of salicylazosulfapyridine to human serum albumin.Acta Pharmacol. Toxicol. 41:401–416 (1977).
R. D. Gray and S. D. Stroupe. Kinetics and mechanism of bilirubin binding to human serum albumin.J. Biol. Chem. 253:4370–4377 (1978).
K. Kramer, K. Thurau, and P. Deetjen. Hämodynamik des Nierenmarks. I. Mitteilung. Cappilläre Passagezeit, Blutvolumen, Durchblutung, Gewebshämatokrit und O2-Verbrauch des Nierenmarks in Situ.Pflugers Arch. Ges. Physiol. 270:251–269 (1960).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jansen, J.A. Influence of plasma protein binding kinetics on hepatic clearance assessed from a “tube” model and a “well-stirred” model. Journal of Pharmacokinetics and Biopharmaceutics 9, 15–26 (1981). https://doi.org/10.1007/BF01059340
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059340