Summary
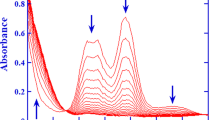
The kinetics of oxidation of [CrIII(nta)(H2O)2] (nta is nitrilotriacetate) by periodate obey the rate law d[CrVI]/dt=(k2[IO −4 ]+k3[IO −4 ]2)[CrIII(nta)(H2O)2] under fixed conditions. The activation parameters are reported and we propose that electron-transfer proceeds via an inner-sphere mechanism.
Similar content being viewed by others
References
G. J. Buist, in C. H. Bamford and C. F. H. Tipper (Eds),Comprehensive Chemical Kinetics, Vol. 6, Elsevier, Amsterdam, 1972, p. 435.
A. Indelli, F. Ferranti, and F. Secco,J. Phys. Chem.,70, 631 (1966).
F. R. El-Eziri and Y. Sulfab,Inorg. Chim. Acta,24, 15 (1977).
A. Y. Kassim and Y. Sulfab,Inorg. Chim. Acta,24, 247 (1977).
A. A. Abdel-Khalek and Y. Sulfab,J. Inorg. Nucl. Chem.,43, 3257 (1981).
M. A. Hussein, A. A. Abdel-Khalek and Y. Sulfab,J. Chem. Soc., Dalton Trans., 317 (1983).
A. Y. Kassim and Y. Sulfab,Inorg. Chem.,20, 506 (1981).
N. A. Al-Jallal and Y. Sulfab,Transition Met. Chem.,8, 51 (1983).
M. C. R. Symon,J. Chem. Soc., 2794 (1955).
G. Den Boef and B. C. Poeder,Anal. Chim. Acta,30, 261 (1964).
G. Schwazenbach and W. Biederman,Helv. Chim. Acta,31, 331 (1948).
I. Hadinec, L. Jensovsky, A. Linek and V. Synecek,Naturwiss,47, 377 (1960).
R. G. Wilkins,The Study of Kinetics and Mechanism of Reactions of Transition Metal Complexes, Allyn and Bacon, Boston, 1974, p. 100.
D. J. B. Galliford and J. M. Ottaway,Analyst. 91, 415 (1972).
B. J. Buist, W. C. P. Hipperson and J. D. Lewis,J. Chem. Soc. (A), 307 (1969).
T. W. Kallen, M. J. Root and K. A. Schroeder,Inorg. Chem.,18, 3318 (1979).
D. D. Perrin,Stability Constants of Metal Complexes, Part B, IUPAC Chemical Data Series No. 22, Pergamon Press, Oxford, p. 420.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abdel-Khalek, A.A., Elsemongy, M.M. Kinetics of the oxidation of diaqua(nitrilotriacetato)-chromium(III) by periodate in aqueous solutions. Transition Met Chem 14, 206–208 (1989). https://doi.org/10.1007/BF01043796
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01043796