Abstract
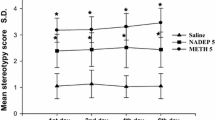
p-Chloroamphetamine (PCA) is a useful pharmacologic tool for selectively increasing brain serotonin function acutely by release of serotonin into the synaptic cleft. PCA produces behavioral, neurochemical and neuroendocrine effects believed due to serotonin release after doses in the range of 0.5–5 mg/kg. At higher doses and at longer times, PCA causes depletion of brain serotonin. The mechanisms of this depletion are not well understood but require the serotonin uptake carrier. Antagonism of PCA-induced depletion of brain serotonin is a useful means of assessing the ability of a compound to block the serotonin uptake carrier on brain serotonin neurons. PCA can also be used as a neurotoxic agent to deplete brain serotonin in functional studies, apparently by destroying some serotonergic nerve terminals. Used in this way, PCA has an advantage over 5,6- and 5,7-dihydroxytryptamines in being effective by systemic injection, and it affects brain serotonergic projections with a different neuroanatomic specificity than the dihydroxytryptamines.
Similar content being viewed by others
References
Pletscher, A., Burkhard, W. P., Bruderer, H., and Gey, K. F. 1963. Decrease of cerebral 5-hydroxytryptamine and 5-hydroxy-indoleacetic acid by an arylalkylamine. Life Sci. 11:828–833.
Owen, J. E., Jr. 1963. Psychopharmacological studies of some 1-(chlorophenyl)-2-aminopropanes. I. Effects on appetite-controlled behavior. J. Pharm. Sci. 52:679–683.
Owen, J. E., Jr. 1963. Psychopharmacological studies of some 1-(chlorophenyl)-2-aminopropanes. II. Effects on avoidance and discrimination behavior. J. Pharm. Sci. 52:684–688.
Fuller, R. W., Hines, C. W., and Mills, J. 1964. Lowering of rat brain serotonin levels by arylalkylamines. Fed. Proc. 23:146.
Fuller, R. W., Hines, C. W., and Mills, J. 1965. Lowering of brain serotonin level by chloramphetamines. Biochem. Pharmacol. 14:483–488.
Sanders-Bush, E., and Sulser, F. 1970.p-Chloroamphetamine: In vivo investigations on the mechanism of action of the selective depletion of cerebral serotonin. J. Pharmacol. Exp. Ther. 175:419–426.
Sanders-Bush, E., Bushing, J. A., and Sulser, F. 1972.p-Chloroamphetamine—Inhibition of cerebral tryptophan hydroxylase. Biochem. Pharmacol. 21:1501–1510.
Sanders-Bush, E., Bushing, J. A., and Sulser, F. 1972. Long-term effects ofp-chloroamphetamine on tryptophan hydroxylase activity and on the levels of 5-hydroxytryptamine and 5-hydroxy-indoleacetic acid in brain. Eur. J. Pharmacol. 20:385–388.
Fuller, R. W., and Baker, J. C. 1977. The role of metabolic N-dealkylation in the action of p-chloroamphetamine and related drugs on brain 5-hydroxytryptamine. J. Pharm. Pharmacol. 29:561–562.
Fuller, R. W., Snoddy, H. D., and Perry, K. W. 1987.p-Chloroamphetamine formation responsible for long-term depletion of brain serotonin after N-cyclopropyl-p-chloroamphetamine injection in rats. Life Sci. 40:1921–1927.
Huang, X., Johnson, M. P., Oberlender, R., Nash, J. F., and Nichols, D. E. 1990. Acute and chronic effects of the α-ethyl homologue ofp-chloroamphetamine (PCA). Soc. Neurosci. Abstr. 16:1033.
Fuller, R. W., Schaffer, R. J., Roush, B. W., and Molloy, B. B. 1972. Drug disposition as a factor in the lowering of brain serotonin by chloroamphetamines in the rat. Biochem. Pharmacol. 21:1413–1417.
Pletscher, A., Bartholini, G., Bruderer, H., Burkhard, W. P., and Gey, K. F. 1964. Chlorinated arylalkylamines affecting the cerebral metabolism of 5-hydroxytryptamine. J. Pharmacol. Exp. Ther. 145:344–350.
Fuller, R. W., Snoddy, H. D., and Molloy, B. B. 1973. Effect of β,β-difluoro substitution on the disposition and pharmacological effects of 4-chloroamphetamine in rats. J. Pharmacol. Exp. Ther. 184:278–284.
Fuller, R. W., Snoddy, H. D., Roush, B. W., and Molloy, B. B. 1973. Further structure-activity studies on the lowering of brain 5-hydroxyindoles by 4-chloroamphetamine. Neuropharmacology 12:33–42.
Fuller, R. W., and Perry, K. W. 1983. Decreased accumulation of brain 5-hydroxytryptophan after decarboxylase inhibition in rats treated with fenfluramine, norfenfluramine orp-chloroamphetamine. J. Pharm. Pharmacol. 35:597–598.
Gallagher, D. W., and Sanders-Bush, E. 1973. In vivo measurement of the release of 5-hydroxytryptamine (5HT) from the hippocampus of the rat: effect of Ro 4-1284, pargyline andp-chloroamphetamine (PCA). Fed. Proc. 32:303.
Marsden, C. A., Conti, J., Strope, E., Curzon, G., and Adams, R. N. 1979. Monitoring 5-hydroxytryptamine release in the brain of the freely moving unanaesthetized rat using in vivo voltammetry. Brain Res. 171:85–99.
Blier, P., Serrano, A., and Scatton, B. 1990. Differential responsiveness of the rat dorsal and median raphe 5-HT systems to 5-HT1-receptor agonists and p-chloroamphetamine. Synapse 5:120–133.
Hutson, P. H., and Curzon, G. 1989. Concurrent determination of effects ofp-chloroamphetamine on central extracellular 5-hydroxytryptamine concentration and behaviour. Brit. J. Pharmacol. 96:801–806.
Adell, A., Sarna, G. W., Hutson, P. H., and Curzon, G. 1989. An in vivo dialysis and behavioural study of the release of 5-HT byp-chloroamphetamine in reserpine-treated rats. Brit. J. Pharmacol. 97:206–212.
Growdon, J. H. 1977. Postural changes, tremor, and myoclonus in the rat immediately following injections ofp-chloroamphetamine. Neurology 27:1074–1077.
Trulson, M. E., and Jacobs, B. L. 1976. Behavioral evidence for the rapid release of CNS serotonin by PCA and fenfluramine. Eur. J. Pharmacol. 36:149–154.
Davis, M., and Sheard, M. H. 1976.p-Chloroamphetamine (PCA): Acute and chronic effects on habituation and sensitization of the acoustic startle response in rats. Eur. J. Pharmacol. 35:261–273.
Zemlan, F. P., Trulson, M. E., Howell, R., and Hoebel, B. G. 1977. Influence ofp-chloroamphetamine on female sexual reflexes and brain monoamine levels. Brain Res. 123:347–356.
Orikasa, S., and Sloley, B. D. 1988. Effects of 5,7-dihydroxytryptamine and 6-hydroxydopamine on head-twitch response induced by serotonin,p-chloroamphetamine, and tryptamine in mice. Psychopharmacology 95:124–131.
Fuller, R. W., and Snoddy, H. D. 1980. Effect of serotonin-releasing drugs on serum corticosterone concentration in rats. Neuroendocrinology 31:96–100.
Fuller, R. W., Snoddy, H. D., and Clemens, J. A. 1980. Elevation of serum prolactin acutely after administration ofp-chloroamphetamine in rats. Endocr. Res. Commun. 7:77–85.
Van de Kar, L. D., Wilkinson, C. W., and Ganong, W. F. 1981. Pharmacological evidence for a role of brain serotonin in the maintenance of plasma renin activity in unanesthetized rats. J. Pharmacol. Exp. Ther. 219:85–90.
Frey, H.-H. 1970.p-Chloroamphetamine — similarities and dissimilarities to amphetamine. Pages 343–347,in Costa, E. and Garattini, S. (eds.), Amphetamines and Related Compounds, Raven Press, New York.
Fuller, R. W., and Molloy, B. B. 1974. Recent studies with 4-chloroamphetamine and some analogues. Adv. Biochem. Psychopharmacol. 10:195–205.
Sanders-Bush, E., and Steranka, L. R. 1978. Immediate and long-term effects ofp-chloroamphetamine on brain amines. Ann. N. Y. Acad. Sci. 305:208–221.
Hashimoto, K., and Goromaru, T. 1990. High-affinity [3H]6-nitroquipazine binding sites in rat brain. Eur. J. Pharmacol. 180:273–281.
Mamounas, L. A., and Molliver, M. E. 1988. Evidence for dual serotonergic projections to neocortex: axons from the dorsal and median raphe nuclei are differentially vulnerable to the neurotoxinp-chloroamphetamine (PCA). Exp. Neurol. 102:23–36.
Commins, D. L., Axt, K. J., Vosmer, G., and Seiden, L. S. 1987. Endogenously produced 5,6-dihydroxytryptamine may mediate the neurotoxic effects of para-chloroamphetamine. Brain Res. 419:253–261.
Fritschy, J. M., Lyons, W. E., Molliver, M. E., and Grzanna, R. 1988. Neurotoxic effects ofp-chloroamphetamine on the serotonergic innervation of the trigeminal motor nucleus: a retrograde transport study. Brain Res. 473:261–270.
Meek, J. L., Fuxe, K., and Carlsson, A. 1971. Blockade ofp-chloroamphetamine induced 5-hydroxytryptamine depletion by chlorimipramine, chlorpheniramine and meperidine. Biochem. Pharmacol. 20:707–709.
Fuller, R. W. 1980. Mechanism by which uptake inhibitors antagonizep-chloroamphetamine-induced depletion of brain serotonin. Neurochem. Res. 5:241–245.
Fuller, R. W., Perry, K. W., and Molloy, B. B. 1975. Reversible and irreversible phases of serotonin depletion by 4-chloroamphetamine. Eur. J. Pharmacol 33:119–124.
Fuller, R. W., Snoddy, H. D., Perry, K. W., Bymaster, F. P., and Wong, D. T. 1978. Importance of duration of drug action in the antagonism ofp-chloroamphetamine depletion of brain serotonin—comparison of fluoxetine and chlorimipramine. Biochem. Pharmacol. 27:193–198.
Berger, U. V., Grzanna, R., and Molliver, M. E. 1989. Depletion of serotonin usingp-chloroamphetamine (PCA) in the brain. Exp. Neurol. 103:111–115.
Axt, K. J., and Seiden, L. S. 1990. α-Methyl-p-tyrosine partially attenuatesp-chloroamphetamine-induced 5-hydroxytryptamine depletions in the rat brain. Pharmacol. Biochem. Behav. 35:996–997.
Sharp, T., Zetterstrom, T., Christmanson, L., and Ungerstedt, U. 1986.p-Chloroamphetamine releases both serotonin and dopamine into rat brain dialysates in vivo. Neurosci. Lett. 72:320–324.
Steranka, L. R., and Rhind, A. W. 1987. Effect of cysteine on the persistent depletion of brain monoamines by amphetamine,p-chloroamphetamine and MPTP. Eur. J. Pharmacol. 133:191–197.
Invernizzi, R., Fracasso, C., Caccia, S. Di Clemente, A., Garattini, S., and Samanin, R. 1989. Effect of L-cysteine on the long-term depletion of brain indoles caused byp-chloroamphetamine and d-fenfluramine in rats. Relation to brain drug concentrations. Eur. J. Pharmacol. 163:77–83.
Steranka, L. R., and Sanders-Bush, E. 1978. Long-term effects on continuous exposure top-chloroamphetamine: effects of inducers and inhibitors of drug metabolism. J. Pharmacol. Exp. Ther. 206:460–467.
Fuller, R. W., Perry, K. W., Baker, J. C., Parli, C. J., Lee, N., Day, W. A., and Molloy, B. B. 1974. Comparison of the oxime and the hydroxylamine derivatives of 4-chloroamphetamine as depletors of brain 5-hydroxyindoles. Biochem. Pharmacol. 23:3267–3272.
Miller, K. J., Anderholm, D. C., and Ames, M. M. 1986. Metabolic activation of the serotonergic neurotoxin para-chloroamphetamine to chemically reactive intermediates by hepatic and brain microsomal preparations. Biochem. Pharmacol. 35:1737–1742.
Berger, U. V., Molliver, M. E., and Grzanna, R. 1990. Unlike systemic administration ofp-chloroamphetamine, direct intracerebral injection does not produce degeneration of 5-HT axons. Exp. Neurol. 109:257–268.
Vorhees, C. V., Schaefer, G. J., and Barrett, R. J. 1975.p-Chloroamphetamine: behavioral effects of reduced cerebral serotonin in rats. Pharmacol. Biochem. Behav. 3:279–284.
Grabowska, M., and Michaluk, J. 1974. On the role of serotonin in apomorphine-induced locomotor stimulation in rats. Pharmacol. Biochem. Behav. 2:263–266.
Ogren, S.-O., Kohler, C., Ross, S. G., and Srebro, B. 1976. 5-Hydroxytryptamine depletion and avoidance acquisition in the rat. Antagonism of the long-term effects ofp-chloroamphetamine with a selective inhibitor of 5-hydroxytryptamine uptake. Neurosci. Lett. 3:341–347.
Altman, H. J., Ogren, S. O., Berman, R. F., and Normile, H. J. 1989. The effects ofp-chloroamphetamine, a depletor of brain serotonin, on the performance of rats in two types of positively reinforced complex spatial discrimination tasks. Behav. Neural Biol. 52:131–144.
Gob, R., Kollner, U., Kollner, O., and Klingberg, F. 1987. Early postnatal development of open field behaviour is changed by single doses of fenfluramine orp-chloroamphetamine. Biomed. Biochim. Acta 46:189–198.
Kollner, O., Kollner, U., Gob, R., and Klingberg, F. 1987. Postnatal application ofp-chloroamphetamine or fenfluramine reduces response selection during early ontogenetic development of rat avoidance behaviour. Biomed. Biochim. Acta 47:997–1005.
Nabeshima, T., Yamaguchi, K., Ishikawa, K., Furukawa, H., and Kameyama, T. 1989. Potentiation of phencyclidine and serotonin agonist-induced behaviors after administration ofp-chloroamphetamine in rats. Res. Commun. Subst. Abuse 10:37–51.
Nabeshima, T., Yamaguchi, K., Ishikawa, K., Furukawa, H., and Kameyama, T. 1987. Potentiation in phencyclidine-induced serotonin-mediated behaviors after intracerebroventricular administration of 5,7-dihydroxytryptamine in rats. J. Pharmacol. Exp. Ther. 243:1139–1146.
Hunskaar, S., Berge, O. G., Broch, O. J., and Hole, K. 1986. Lesions of the ascending serotonergic pathways and antinociceptive effects after systemic administration ofp-chloroamphetamine in mice. Pharmacol. Biochem. Behav. 24:709–714.
Kutscher, C. L., and Yamamoto, B. K. 1979. A frequency analysis of behavior components of the serotonin syndrome produced byp-chloroamphetamine. Pharmacol. Biochem. Behav. 11:611–616.
Marsden, C. A. 1979. Long term effects ofp-chloroamphetamine on hippocampal 5-hydroxytryptamine release. Brit. J. Pharmacol. 66:120P.
Archer, T., Ogren, S.-O., and Ross, S. B. 1982. Serotonin involvement in aversive conditioning: reversal of the fear retention deficit by long-termp-chloroamphetamine but notp-chlorophenylalanine. Neurosci. Lett. 34:75–82.
Fuller, R. W. 1978. Neurochemical effects of serotonin neurotoxins: An introduction. Ann. N. Y. Acad. Sci. 305:178–181.
Sanders-Bush, E., Bushing, J. A., and Sulser, F. 1975. Long-term effects ofp-chloroamphetamine and related drugs on central serotonergic mechanisms. J. Pharmacol. Exp. Ther. 192:33–41.
Alesci, R., and Bagnoli, P. 1988. Endogenous levels of serotonin and 5-hydroxyindoleacetic acid in specific areas of the pigeon CNS: effects of serotonin neurotoxins. Brain Res. 450:259–271.
Bjorkum, A. A., and Berge, O. G. 1988. The relative contribution of ascending and descending pathways inp-chloroamphetamine-induced antinociception. Pharmacol. Biochem. Behav. 31:135–140.
Perrone, M. H., Luttinger, D., Hamel, L. T., Fritz, P. M., Ferraino, R., and Haubrich, D. R. 1990. In vivo assessment of napamezole, an alpha-2 adrenoceptor antagonist and monoamine reuptake inhibitor. J. Pharmacol. Exp. Ther. 254:476–483.
Thomas, D. R., Nelson, D. R., and Johnson, A. M. 1987. Biochemical effects of the antidepressant paroxetine, a specific 5-hydroxytryptamine uptake inhibitor. Psychopharmacology 93:193–200.
Steranka, L. R., and Sanders-Bush, E. 1978. Long-term effects of continuous exposure top-chloroamphetamine on central serotonergic mechanisms in mice. Biochem. Pharmacol. 27:2033–2037.
Eide, P. K., Hole, K., Berge, O. G., and Broch, O. J. 1988. 5-HT depletion with 5,7-DHT, PCA and PCPA in mice: differential effects on the sensitivity to 5-MeODMT, 8-OH-DPAT and 5-HTP as measured by two nociceptive tests. Brain Res. 440:42–52.
Hunskaar, S., Fasmer, O. B., Broch, O. J., and Hole, K. 1987. Involvement of central serotonergic pathways in nefopam-induced antinociception. Eur. J. Pharmacol. 138:77–82.
Heinsbroek, R. P., Feenstra, M. G., Boon, P., Van Haaren, F., and Van de Poll, N. E. 1988. Sex differences in passive avoidance depend on the integrity of the central serotonergic system. Pharmacol. Biochem. Behav. 31:499–503.
Yamaguchi, K., Nabeshima, T., and Kameyama, T. 1986. Potentiation of phencyclidine-induced dopamine-dependent behaviors in rats after pretreatments with serotonin-depletors. J. Pharmacobiodyn. 9:479–489.
Rowland, D., Steele, M., and Moltz, H. 1978. Serotonergic mediation of the suckling-induced release of prolactin in the lactating rat. Neuroendocrinology 26:8–14.
Gazzara, R. A., Takeda, H., Cho, A. K., and Howard, S. G. 1989. Inhibition of dopamine release by methylenedioxymethamphetamine is mediated by serotonin. Eur. J. Pharmacol. 168:209–217.
Dilsaver, S. C., Normile, H. J., and Altman, H. J. 1989. Potentiation of oxotremorine-induced hypothermia by alaproclate in PCA lesioned and non-lesioned rats. Psychopharmacology 97:51–53.
Freo, U., Larson, D. M., Tolliver, T., Rapoport, S. I., and Soncrant, T. T. 1991. Parachloroamphetamine selectively alters regional cerebral metabolic responses to the serotonin agonist metachlorophenylpiperazine in rats. Brain Res. 544:17–25.
Author information
Authors and Affiliations
Additional information
Special issue dedicated to Dr. Morris H. Aprison.
Rights and permissions
About this article
Cite this article
Fuller, R.W. Effects ofp-chloroamphetamine on brain serotonin neurons. Neurochem Res 17, 449–456 (1992). https://doi.org/10.1007/BF00969891
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00969891