Abstract
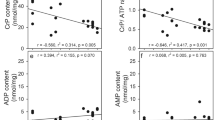
It is often assumed that adenosine transport into brain cells occurs by facilitated diffusion and that the continued net uptake of adenosine depends on its subsequent metabolism, which keeps the intracellular concentration of unmetabolized adenosine low and thus maintains a concentration gradient. If that is the case, inhibition of adenosine metabolism should decrease uptake. We have previously reported a considerable deamination of accumulated adenosine to inosine in primary cultures of cerebral cortical neurons. A relatively specific adenosine deaminase inhibitor, 2′-deoxycoformycin, was used in the present study. In the presence of this drug, the adenosine content (pool size) increased many fold without any decrease in total influx of adenosine. Influx of accumulated adenosine took place against a concentration gradient, demonstrating that a metabolic degradation of accumulated adenosine is not required to drive adenosine uptake. This does not preclude that under normal conditionssome adenosine may get into the cells by diffusion.
Similar content being viewed by others
References
Thampy, G. K., and Barnes, E. M. Jr. 1983. Adenosine transport by primary cultures of neurons from chick embryo brain. J. Neurochem. 40:874–879.
Thampy, G. K., and Barnes, E. M. Jr. 1983. Adenosine transport by cultured glial cells from chick embryo brain. Arch. Biochem. Biophys. 220:340–346.
Geiger, J. D., Johnston, M. E., and Yago, V. 1988. Pharmacological characterization of rapidly accumulated adenosine by dissociated brain cells from adult rat. J. Neurochem. 51:283–291.
Matz, H., and Hertz, L. 1989. Adenosine metabolism of neurons and astrocytes in primary cultures. J. Neurosci. Res., in press.
Phillis, J.W., and Wu, P. H. 1981. The role of adenosine and its nucleotides in central synaptic transmission. Prog. Neurobiol. 16:187–239.
Snyder, S. H. 1985. Adenosine as a neuromodulator. Ann. Rev. Neurosci. 8:103–124.
Agarwal, R. P. 1982. Inhibitors of adenosine deaminase. Pharmac. Ther. 17:399–429.
Henderson, J. F., Brox, L., Zombor, G., Hunting, D. and Lomax, C. A. 1977. Specificity of adenosine deaminase inhibitors. Biochem. Pharmacol. 26:1967–1972.
Hertz, L., Juurlink, B. H. J., and Szuchet, S. 1985. Cell cultures. Vol. 8, Pages 603–661,in Latha, A. (ed.), Handbook of Neurochemistry, 2nd Edition, Plenum Press, New York.
Hertz, E., Yu, A. C. H., Hertz, L., Jurrlink, B. H. J., and Schousboe, A. 1989. Preparation of primary cultures of mouse cortical neurons. Pages 183–186, in Shahar, A., Vernadakis, A., De Vellis, J., and Haber, B. (eds.), A dissection and tissue culture manual for the nervous system. Liss, A.R., New York.
Kuriyama, K., Ohkuma, S., Tomono, S., Hirouchi, M., and Hashimoto, T. 1986. Developmental patterns of GABA-ergic neuron and GABAA receptor: analysis using cerebral cortical neurons in primary cultures. Int. J. Develop. Neurosci. 4, Suppl 1, S43.
Lowry, O. H., Roseborough, N. J., Farr, A. L., and Randall, R. J. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–275.
Webster, D. R., Boston, G. D., and Paton, D. M. 1985. Measurement of adenosine metabolites and metabolism in isolated tissue preparations. J. Pharmacol. Methods 13:339–350.
Khym, J. X. 1975. An analytical system for rapid separation of tissue nucleotides at low pressure on conventional anion exchangers. Clin. Chem. 21:1245–1252.
Schweinsberg, P. D., and Loo, T. L. 1980. Simultaneous analysis of ATP, ADP, AMP, and other purines in human erthrocytes by high-performance liquid chromatography. J. Chromatogr. 181:103–107.
Berl, S., and Clarke, D. D. 1975. Pages xiii-xviiin Berl, S., Clarke, D. D., and Schneider, D. (eds.), Metabolic compartmentation and neurotransmission: relation to brain structure and function. Plenum Press, New York.
Hertz, L. 1978. Kinetics of adenosine uptake into astrocytes. J. Neurochem. 31:55–62.
Bender, A. S., and Hertz, L. 1986. Similarities of adenosine uptake systems in astrocytes and neurons in primary cultures. Neurochem. Res. 11:1507–1524.
Barberis, C., Minn, A., and Gayet, J. 1981. Adenosine transport into guinea-pig synaptosomes. J. Neurochem. 36:347–354.
Banay-Schwartz, M., de Guzman, T., and Lajtha, A. 1980. Nucleotide uptake by slices of mouse brain. J. Neurochem. 35:544–551.
Bender, A. S., Wu, P. H., and Phillis, J. W. 1980. The characterization of [3H] adenosine uptake into rat cerebral cortical synaptosomes. J. Neurochem. 35:629–640.
Bender, A. S., Wu, P. H., and Phillis, J. W. 1981. The rapid uptake and release of [3H]adenosine by rat cerebral cortical synaptosomes. J. Neurochem. 36:651–660.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hertz, L., Matz, H. Inhibition of adenosine deaminase activity reveals an intense active transport of adenosine into neurons in primary cultures. Neurochem Res 14, 755–760 (1989). https://doi.org/10.1007/BF00964954
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00964954