Summary
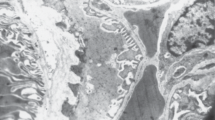
The three-dimensional structure of the neck segment (NS) of the rabbit nephrons was studied by scanning electron microscopy (after fracture, microdissection, or after corrosion nephron casts), and by the air-cast method. The NS was observed at the glomerulotubular junction in 68.5% of all nephrons. In every case the NS appeared as a straight tube with its long axis oriented radially in relation to the glomerulus. Although the external diameter of the NS was smaller than that of the proximal tubule, its luminal diameter was greater. No valve-like structures were observed. Three cell types were observed in the NS: parietal-like, tubule-like, and intermediate. Parietal-like cells showed the same morphology as the parietal cells of the Bowman's capsule. Parietal-like cells constituted the only cell type in 25% of the NS. Tubule-like cells showed morphologic characteristics similar to proximal tubule cells; however, their microvilli were less numerous and exhibited an irregular pattern. Intermediate cells presented an intermediate morphology between tubule-like and parietal cells. In 75% of all NS, the three cellular types were present at the same time. The presence of tubule-like and intermediate cells is interpreted as the result of metaplasic transformation of the parietal cells. Our observations suggest that, in rabbits, the presence of the NS can be explained on the basis of phenotypical lability of the cells located at the glomerulo-tubular junction.
Similar content being viewed by others
References
Andrews PM (1979) The urinary system-Kidney. In: Hodges GM, Hallowes RC (eds) Biomedical research applications of scanning electron microscopy, vol 1. Academic Press, London, pp 273–306
Andrews PM (1981) The presence of proximal tubulelike cells in the kidney parietal epithelium in response to unilateral nephrectomy. Anat Rec 200:61–65
Barberini F, Familiari G, Vittori I, Carpino F, Melis M (1984) Morphological and statistical investigation of the occurrence of “tubule-like cells” in the renal corpuscle of the mouse kidney induced by sex hormones. Renal Physiol 7:227–236
Carpino F, Barberini F, Familiari G, Melis M (1976) Columnar cells of the parietal layer of Bowman's capsule and their relationship with the sexual cycle in normal female mice. Experientia 32:1584–1585
Clothier RH, Worley RTS, Balls M (1978) The structure and ultrastructure of the renal tubule of the urodele amphibian, Amphiuma means. J Anat 127:491–504
Dietert SE (1967) The columnar cells occurring in the parietal layer of Bowman's capsule. Cellular fine structure and protein transport. J Cell Biol 35:435–444
Edwards JG, Schnitter C (1933) The renal unit in the kidney of vertebrates. Am J Anat 53:55–87
Eulderink F (1964) Adenomatoid changes in Bowman's capsule in primary carcinoma of the liver. J Pathol Bacteriol 87:251–254
Evan AP, Dail WG, Dammrose D, Palmer C (1976) Scanning electron microscopy of cell surfaces following removal of extracellular material. Anat Rec 185:433–446
Haensly WE, Granger HJ, Morris AC, Cioffe C (1982) Proximal tubule-like epithelium in Bowman's capsule in spontaneously hypertensive rats. Changes with age. Am J Pathol 107:92–97
Haley DP, Bulger RE (1983) The aging male rat: structure and function of the kidney. Am J Anat 167:1–13
Jackson JD, Smith FG, Litman NN, Yuile CL, Latta H (1962) The Fanconi syndrome with cystinosis: Electron microscopy of renal biopsy specimens from five patients. Am J Med 33:893–910
Kaissling B, Kriz W (1979) Structural analysis of the rabbit kidney. Adv Anat Embryol Cell Biol, Springer, Berlin Heidelberg New York
Lacy ER, Reale E (1985) The elasmobranch kidney. II Sequence and structure of the nephrons. Anat Embryol 173:163–186
Mayer E, Ottolenghi LA (1947) Protrusion of tubular epithelium into the space of Bowman's capsule in kidneys of dogs and cats. Anat Rec 99:477–507
Melis M, Carpino F, Palermo D, Motta P (1974) Scanning and transmission electron microscopic observations on the columnar cells of the parietal layer of Bowman's capsule in normal mice. J Microscopie 19:247–252
Ohtani O (1981) Microcirculation studies by the injection replica method. Biomed Res 2:210–226
Ojeda JL, Garcia-Porrero JA (1982) Structure and development of parietal podocytes in renal glomerular cysts induced in rabbits with methylprednisolone acetate. Lab Invest 47:167–176
Ojeda JL, Garcia-Porrero JA, Ros MA (1984) A new and simple method for studying the microanatomy of nephrons and collecting tubules. Stain Technol 59:37–42
Sakai T (1985) The structure of the kidney from the freshwater telcost Carassius auratus. Anat Embryol 171:31–39
Sakai T, Kawahara K (1983) The structure of the kidney of japanese newts, Triturus (Cynops) pyrrhogaster. Anat Embryol 166:31–52
Schonheyder HC, Maunsbach AB (1975) Ultrastructure of a specialized neck region in the rabbit nephron. Kidney Int 7:145–153
Sperber I (1944) Studies on the mammalian kidney. Zoo Bidrag Uppsala 22:249–431
Von Möllendorff W (1930) Der Exkretionsapparat. In: Handbuch der Mikroskopischen Anatomic des Menschen, vol VII. Springer, Berlin, p 1
Youson JH, McMillan DB (1970) The opisthonephric kidney of the sea lamprey of the Great Lakes, Petromyzon marinus L. II Neck and proximal segments of the tubular nephron. Am J Anat 127:233–258
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ojeda, J.L., Icardo, J.M. A scanning electron microscope study of the neck segment of the rabbit nephron. Anat Embryol 184, 605–610 (1991). https://doi.org/10.1007/BF00942582
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00942582