Abstract
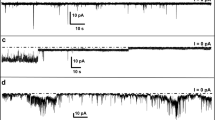
The Alzheimer's Disease (AD) amyloid protein (AßP[1-40]) forms cation selective channels when incorporated into planar lipid bilayers by fusion with liposomes containing the peptide. Since the peptide has been proposed to occurin vivo in both membrane-bound and soluble forms, we also tested the possibility of direct incorporation of the soluble AßP[1-40] into the membrane. We found the peptide can also form similar channels in acidic phospholipid bilayers formed at the tip of a patch pipet, as well as in the planar lipid bilayer system. As in the case of liposome mediated incorporation, the AßP[1-40]-channel in the solvent-free membrane patch exhibits multiple cation selectivity (Cs+>Li+>Ca2+≥K+) and sensitivity to tromethamine. The fact that equivalentAßP[1-40] amyloid channels can be detected by two different methods thus provides additional validation of our original observation. Further studies with aßP-channels incorporated into planar lipid bilayers from the liposome complex have also revealed that the channel activity can express spontaneous transitions to a much higher range of conductances between 400 and 4000 pS. Under these conditions, the amyloid channel continues to be cation selective but loses its tromethamine sensitivity. By contrast, amyloid channels were insensitive to nitrendipine at either conductance range. We calculate that if such channels were expressed in cells, the ensuing ion fluxes down their electrochemical potential gradients would disrupt cellular homeostasis. We therefore interpret these data as providing further support for our ß-amyloid Ca2+-channel hypothesis for neuronal death in Alzheimer's Disease.
Similar content being viewed by others
References
Reisberg B, (ed): Alzheimer's disease, The Free Press, New York, 1983
Glenner GG, Wong CW: Alzheimer's disease: Initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun 120: 885–890, 1984
Masters CL, Simms G, Weinman NA, Multhaup G, McDonald BI, Beyreuther K: Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc Natl Acad Sci USA 82: 4245–4249, 1985
Frautschy SA, Baird A, Cole GM: Effects of injected Alzheimer beta-amyloid cores in rat brain. Proc Natl Acad Sci USA 88: 8362–8366, 1991
Neve RL, Dawes LR, Yankner BA, Benowitz LL, Rodriguez W, Higgins GA: Genetics and Biology of the Alzheimer amyloid precursor. Prog Brain Res 86: 257–267, 1990
Selkoe DJ: The molecular pathology of Alzheimer's disease. Neuron 6: 487–498, 1991
Hardy JA, Higgins GA: Alzheimer's disease: the amyloid cascade hypothesis. Science 256: 780–783, 1992
Selkoe DJ: Physiological production of the β-amyloid protein and the mechanism of alzheimer's disease. Trends Neurosci 16(10): 403–409, 1993
Kang J, Lemaire H-G, Unterbeck A, Salbaum JM, Masters CL, Grzeschik K-H, Multhaup G, Beyreuther K, Muller-Hill B: The precursor of Alzheimer's disease amyloid A4 protein resembles a cell-surface receptor. Nature 325: 733–736, 1987
Yankner BA, Dawes LR, Fisher S, Villa-Komaroff L, Oster-Granite ML, Neve RL: Neurotoxicity of a fragment of the amyloid precursor associated with Alzheimer's disease. Science 245: 417–420, 1989
Yankner BA, Caceres A, Duffy LK: Nerve growth factor potentiates the neurotoxicity of β amyloid. Proc Natl Acad Sci USA 87: 9020–9023, 1990
Yankner BA, Duffy LK, Kirschner DA: Neurotrophic and neurotoxic effects of β amyloid protein: reversal by tachykinin neuropeptides. Science 250: 279–282, 1990
Roher AE, Ball MJ, Bhave SV, Wakade AR: β-amyloid protein from Alzheimer's disease brain inhibits sprouting and survival of sympathetic neurons. Biochem Biophys Res Commun 174: 572–579, 1991
Kowall NW, Mckee AC, Yankner BA, Beal MF:In vivo neurotoxicity of beta amyloid [β(1–40)] and the β(25–35) fragment. Neurobiol Aging 13(5): 537–542, 1992
Malouf AT: Effect of beta amyloid peptides on neurons in hippocampal slice cultures: Neurobiol Aging 13(5): 543–551, 1992
Emre M, Geula C, Ransil BJ, Mesulam MM: The acute neurotoxicity and effects upon cholinergic axons of cerebrally injected β-amyloid in the rat brain. Neurobiol Aging 13(5): 553–559, 1992
Yankner BA: Commentary and perspective on studies of beta amyloid neurotoxicity. Neurobiol Aging 13(5): 615–616, 1992
Pike C, Walencewicz AJ, Glabe CG, Cotman CW:In vitro aging of β-amyloid protein causes peptide aggregation and neurotoxicity. Brain Res 563: 311–314, 1991
Pike C, Burdick D, Walencewicz AJ, Glabe CG, Cotman CW: Neurodegeneration induced by β-amyloid peptidesin vitro: the role of peptide assembly state. J Neurosci 13(4): 1676–1687, 1993
Podlisny MB, Stephenson DT, Frosch MP, Lieberburg I, Clemens JA, Selkoe DJ: Synthetic amyloid β-protein fails to produce specific neurotoxicity in monkey cerebral cortex. Neurobiology of aging 13(5): 561–567, 1992
Games D, Khan KM, Soriano FG, Keim PS, Davis DL, Bryani K, Lieberburg I: Lack of alzheimer's pathology after β-amyloid protein injections in rat brain. Neurobiology of aging 13(5): 569–576, 1992
Stein-Behrens B, Adams K, Yeh M, Sapolsky R: Failure of beta-amyloid protein fragment 25–35 to cause hippocampal damage in the rat. Neurobiol Aging 13(5): 577–579, 1992
Cotman CW, Pike CJ, Copani A: β-amyloid neurotoxicity: A discussion ofin vitro findings. Neurobiol Aging 13(5): 587–590, 1992
Simmons MA, Schneider CR: Amyloid β-peptides act directly on single neurons. Neurosci Lett 150: 133–136, 1993
Busciglio J, Yeh J, Yankner BA: β-amyloid neurotoxicity in human cortical culture is not mediated by excitotoxins. J Neurochem 61(4): 1565–1568, 1993
Arispe N, Rojas E, Pollard HB: Alzheimer disease amyloid β protein forms calcium channels in bilayer membranes: Blockade by tromethamine and aluminum. Proc Natl Acad Sci USA 90: 567–571, 1993
Arispe N, Pollard HB, Rojas E: Giant multilevel cation channels formed by alzheimer disease amyloid β-protein [AßP-(1-40)] in bilayer membranes. Proc Natl Acad Sci USA 90: 10573–10577, 1993
Khachaturian ZS: The role of calcium regulation in brain aging:reexamination of a hypothesis. Aging (Milano) 1(1): 17–34, 1989
Mattson MP, Cheng B, Davis D, Bryant K, Lieberburg I, Rydel R: β-amyloid peptides destabilize calcium homeostasis and render human cortical neurons vulnerable to excitotoxicity. J Neurosci 12(2): 376–386, 1992
Mattson MP, Tomaselli KJ, Rydel RE: Calcium-destabilizing and neurodegenerative effects of aggregated β-amyloid peptide are attenuated by basic FGF. Brain Research 621: 35–49, 1993
Mattson MP, Barger SW, Cheng B, Lieberburg I, Smith-Swintosky VL, Ryder R: β-amyloid precursor protein metabolites and loss of neuronal Ca2+ homeostasis in alzheimer's disease. Trends Neurosci 16: 409–414, 1993
Choi DW: Calcium-mediated neurotoxicity: relationship to specific channel types and role in ischemic damage. Trends Neurosci 11(10): 465–469, 1988
Miller RJ: The control of neuronal Ca++ homeostasis. Progress in Neurobiol 37: 255–285, 1991
Burdick D, Soreghan B, Kwon M, Kosmoski J, Knauer M, Henschen A, Yates J, Cotman C, Gable C: Assembly and aggregation properties of synthetic alzheimer's A4/β amyloid peptide analogs. J Biol Chem 267: 546–554, 1992
Barrow CJ, Yasuda A, Kenny PTM, Zagorski G: Solution conformations and aggregational properties of synthetic amyloid β-peptides of Alzheimer's Disease. Analysis of circular dichroism spectra. J Mol Biol 225: 1075–1093, 1992
Hilbich C, Kisters-woike B, Reed J, Master C, Beyreuther K: Aggregation and secondary structure of synthetic amyloid βA4 peptides of Alzheimer's disease. J Mol Biol 218: 149–163, 1991
Mason RP, Shoemaker WJ, Shajenko L, Herbette LG: X-ray diffraction analysis of brain lipid membrane structure in alzheimer's disease and β-amyloid peptide interactions. Ann NY Acad Sci 695: 231–234, 1993
Jarret JT, Lansbury T Jr: Seeding ‘one dimensional crystalization’ of amyloid: A pathogenic mechanism in Alzheimer's disease and scrapie. Cell 73: 1055–1058, 1993
Boheim G: Statistical analysis of alamethicin channels in black lipid membranes. J Memb Biol 19: 277–303, 1974
Boheim G, Kolb H-A: Analysis of the multi-pore system of alamethicin in a lipid membrane. J Memb Biol 38: 99–150, 1978
Shoji M, Golde TE, Ghiso J, Cheung TT, Estus S, Shaffer LM, Cai X-D, McKay DM, Tintner R, Frangione B, Younkin SG: Production of the alzheimer's amyloid β protein by normal processing. Science 258: 126–129, 1992
Seubert P, Oltersdorf T, Lee MG, Barbour R, Blomquist C, Davis DL, Bryant K, Fritz LC, Galasko D, Thai LJ, Lieberburg I, Schenk DB: Secretion of β-amyloid precursor protein cleaved at the amino terminus of the β-amyloid peptide. Nature 361: 260–263, 1993
Haass C, Selkoe J: Cellular processing of β-amyloid precursor protein and the genesis of amyloid b-peptide. Cell 75: 1039–1043, 1993
Fraser PE, Nguyen JT, Surewicz WK, Kirschner DA: pH-dependent structural transitions of alzheimer amyloid peptides. Biophys J 60: 1190–1201, 1992
Inouye H, Fraser PE, Kirschner DA: Structure of β-crystallite assemblies formed by alzheimer β-amyloid protein analogues: analysis oy x-ray diffraction. Biophys J 64: 502–519, 1993
Durell RS, Guy RJ, Arispe N, Rojas E, Pollard HB: Theoretical models of the ion channel structure of amyloid β-protein. Biophys.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arispe, N., Pollard, H.B. & Rojas, E. β-amyloid Ca2+-channel hypothesis for neuronal death in Alzheimer Disease. Mol Cell Biochem 140, 119–125 (1994). https://doi.org/10.1007/BF00926750
Issue Date:
DOI: https://doi.org/10.1007/BF00926750