Abstract
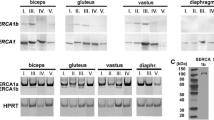
The Western blotting technique was used to detect parvalbumin and S-100 protein in muscles from 10 Duchenne muscular dystrophy (DD) patients, 13 patients with other muscle diseases and 5 age-matched healthy subjects. DD muscles were found to contain decreased amounts of parvalbumin and the S-100 protein. The parvalbumin level did not relate to the age of the patients and the stage of the disease. The S-100 protein decreased progressively with the age of the patients. In a very advanced DD case the S-100 protein was present in trace amounts. In other primary myopathies, including Becker dystrophy, and neurogenic muscular atrophy both parvalbumin and S-100 protein levels were similar to that observed in healthy subjects. The decrease in the amount of both calcium binding proteins may contribute to the elevation of free intracellular Ca2+ level in the sarcoplasm of dystrophic muscle and would result in abnormalities in processes regulated by these proteins. The mechanism(s) responsible for the decrease of parvalbumin and S-100 protein in DD muscles are discussed.
Similar content being viewed by others
References
Bertorini TE, Bhattacharya SK, Palmieri GM, Chesney CM, Pifer D, Baker B (1982) Muscle calcium and magnesium content in Duchenne muscular dystrophy. Neurology 32: 1088–1092
Bertorini TE, Cornelio F, Bhattacharya SK, Palmieri GMA, Donas I, Dworzak F, Brambieri B (1984) Calcium and magnesium content in fetuses at risk and prenecrotic Duchenne muscular dystrophy. Neurology 34: 1436–1440
Blum HE, Lehky P, Kohler L, SteinEA, Fischer EH (1977) Comparative properties of vertebrate parvalbumins. J Biol Chem 252: 2834–2838
Bonilla E, Samitt CE, Miranda AF, Hays AP, Salviati G, Di Mauro S, Kunkel LM, Hoffman EP, Rowland LP (1988) Duchenne muscular dystrophy: deficiency of dystrophin at the cell surface. Cell 54: 447–452
Celio MR, Heizmann CW (1982) Calcium-binding protein parvalbumin is associated with fast contracting muscle fibres. Nature 297: 504–506
Cullen MJ, Fulthorpe JJ (1975) Stages in fibre breakdown in Duchenne muscular dystrophy. J Neurol Sci 24: 179–200
Donato R (1986) S-100 proteins. Cell Calcium 7: 123–145
Dubowitz V, Brooke MH (1973) Muscle biopsy: a modern approach. Saunders, London
Emery AEH, Burt D (1980) Intracellular calcium and pathogenesis and prenatal diagnosis of Duchenne muscular dystrophy. BMJ 1: 1355–1357
Endo E, Tanaka T, Isobe T, Kasai H, Okuyama T, Hidaka H (1981) Calcium dependent affinity chromatography of S-100 and calmodulin on calmodulin antagonist coupled Sepharose. J Biol Chem 256: 12485–12489
Engel AG, Banker BQ (1986) Myology. Basic and clinical. Mc Graw-Hill, New York
Gailly PH, Hermans E, Octave JN, Gillis JM (1993) Specific increase of genetic expression of parvalbumin in fast skeletal muscles of mdx mice. FEBS Lett 326: 272–274
Giometti CS, Bárány M, Danon MJ, Anderson NG (1980) Muscle protein analysis. II. Two-dimensional electrophoresis of normal and diseased human muscle. Clin Chem 28: 1152–1156
Green HJ, Klug GA, Reichmann H, Seedorf V, Wiehrer W, Pette D (1984) Exercise-induced fibre type transitions with regard to myosin, parvalbumin, and sarcoplasmic reticulum in muscles of the rat. Pflügers Arch 400: 432–438
Haiech J, Derancourt J, Pechere JF, Demaille JG (1979) Magnesium and calcium binding to parvalbumin: evidence for differences between parvalbumins and explanation of their relaxing factor. Biochemistry 18: 2752–2758
Hoffman EP, Brown RH, Kunkel LM (1987) Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 51: 919–928
Jockusch H, Friedrich G, Zippel M (1990) Serum parvalbumin, an indicator of muscle disease in murine dystrophy and myotonia. Muscle Nerve 13: 551–555
Klug G, Wiehrer W, Reichmann H, Leberer E, Pette D (1983) Relationship between early alternations in parvalbumins, sarcoplasmic reticulum and metabolic enzymes in chronically stimulated fast twitch muscle. Pflügers Arch 399: 280–284
Klug G, Reichman H, Pette D (1985) Decreased parvalbumin contents in skeletal muscles of C57BL/6J(dydJ/dy2J) dystrophic mice. Muscle Nerve 8: 576–579
Laemmli UH (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Leberer E, Pette D (1986) Neural regulation of parvalbumin expression in mammalian skeletal muscle. Biochem 1235: 67–73
Leberer E, Pette D (1986) Immunochemical quantification of sarcoplasmic reticulum Ca-ATP-ase, of calse-questrin and parvalbumin in rabbit skeletal muscles of defined fiber composition. Eur J Biochem 156: 489–496
Leberer E, Seedorf V, Pette D (1986) Neural control of gene expression in skeletal muscle. Calcium-sequestring proteins in developing and chronically stimulated rabbit skeletal muscles. Biochem 1239: 295–300
Lowry OH, Rosebrough NJ, Farr AE, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Maunder CA, Yarom R, Dubowitz V (1977) Electron microscope x-ray microanalysis of normal and diseased human muscle. J Neurol Sci 33: 323–334
Maunder-Sewry CA, Dubowitz V (1979) Myonuclear calcium in carriers of Duchenne muscular dystrophy: an x-ray microanalysis study. J Neurol Sci 42:337–347
Maunder-Sewry CA, Gorodetsky R, Yarom R, Dubowitz V (1980) Element analysis of skeletal muscle in Duchenne muscular dystrophy using xray fluorescence spectroscopy. Muscle Nerve 3: 502–508
Mokri B, Engel AG (1975) Duchenne dystrophy: electron microscopic findings pointing to a basic or early abnormality in the plasma membrane of the muscle fiber. Neurology 25: 1111–1120
Nicholson LVB, Davison K, Falkous G, Harwood C, O'Donnel E, Slater CR, Harris JB (1989) Dystrophin in skeletal muscle. Western blot analysis using a monoclonal antibody. J Neurol Sci 94: 125–136
Niebój-Dobosz I (1981) Biochemical characteristics of progressive muscular dystrophy (in Polish). PhD Thesis, Medical Academy, Warsaw
Niebrój-Dobosz I, Kornguth S, Schutta HS, Siegel FL (1989) Elevated calmodulin levels and reduced calmodulin-stimulated calcium-ATPase in Duchenne progressive muscular dystrophy. Neurology 39: 1610–1614
Niebrój-Dobosz I, Kornguth S, Schutta HS, Siegel FL, Hausmanowa-Petrusewicz I (1989) Proteins of muscle subcellular fractions in Duchenne's progressive muscular dystrophy stained with “Stains-all” cationic carbocyanine dye and with Coomassie Blue. Muscle Nerve 12: 273–280
Peterson GL (1977) A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem 83: 346–356
Potter JD, Dedman JR, Means AR (1977) Ca2+-dependent regulation of cyclic AMP phosphodiesterase by parvalbumin. J Biol Chem 252: 5609–5611
Rowland LP (1980) Biochemistry of muscle membranes in Duchenne muscular dystrophy. Muscle Nerve 3: 3–20
Sano M, Yokota T, EndoT, Tsukagoshi H (1990) A developmental change in the content of parvalbumin in normal and dystrophic mouse (mdx) muscle. J Neurol Sci 97: 261–272
Sugita H, Katagiri T, Shimuzu T, Toyokura Y (1973) Studies on the structural proteins in various neuromuscular diseases. In: Kakulas BA (ed) Basic research in myology, part 1. Excerpta Medica, Amsterdam/American Elsevier, New York, pp 291–297
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedures and some applications. Proc Natl Acad Sci USA 76: 4350–4354
Wnuk W, Cox JA, Stein EA (1982) Parvalbumins and other soluble high affinity calcium-binding proteins from muscle. In: Cheung WY (ed) Calcium and cell function. Academic Press, New York, pp 243–278
Wrogemann K, Jacobson BE, Blanchaer MC (1973) The mechanism of a calcium-associated defect of oxidative phosphorylation in progressive muscular dystrophy. Arch Biochem Biophys 159: 267–278
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Niebrój-Dobosz, I., Łukasiuk, M. & Niebrój-Dobosz, I. Immunoblot analysis of sarcoplasmic calcium binding proteins in Duchenne muscular dystrophy. J Neurol 242, 82–86 (1995). https://doi.org/10.1007/BF00887821
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00887821