Abstract
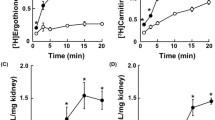
The uptake of the β-amino acid taurine by rat renal brush border membrane vesicles (BBMV) adapts to changes in dietary sulfur amino acid intake. Initial rateV max “upregulates” after ingestion of a low methionine and taurine diet (LTD) and “downregulates” after a high taurine diet (HTD). This is reflected in vivo by hypotaurinuria after a LTD (90% reduction in excretion) and an 18-fold increase in urine taurine after a HTD. This study was performed to determine whether taurine efflux from BBMV is adaptively regulated by external taurine concentration or by diet. Vesicles were preloaded with varying concentrations of radiolabelled and unlabelled taurine and a 150 mM concentration of various salts. Efflux conditions were: taurine and 150 mM salt inside and 150 mM salt outside. The efflux of five concentrations of taurine (10–500 μM) was linear over 6 min, reached equilibrium by 21 min, and was dependent upon intravesicular taurine content. The kinetic characteristics of efflux (E) were significantly different from influx (I):K m=109.8±5.8 (E) versus 23.6±4.2 (I),P<0.001 [time of linearity=360 s (E) vs. 20 s (I)]. Efflux of taurine was dependent on the presence of both sodium and chloride in the system, but neither external taurine content (0,100 μM, 1,000 μM) nor external β-alanine altered initial efflux. Feeding rats a normal diet, LTD, or fasting altered taurine uptake but not efflux. Efflux does not appear to play a role in the adaptive regulation of taurine transport found in all mammalian species.
Similar content being viewed by others
References
Friedman AL, Albright PW, Gusowski N, Padilla M, Chesney RW (1983) Renal adaptation to alteration in dietary amino acid intake. Am J Physiol 245:F159-F166
Chesney RW, Gusowski N, Friedman AL (1983) Renal adaptation to altered dietary sulfur amino acid intake occurs at the luminal brush border membrane. Kidney Int 24:588–594
Chesney RW, Gusowski N, Dabbagh S (1985) Renal cortex taurine content regulates renal adaptive response to altered dietary intake of sulfur amino acids. J Clin Invest 76:2213–2221
Chesney RW, Gusowski N, Dabbagh S (1987) Studies on renal adaptation to altered dietary amino acid intake: reduced renal taurine cortex content increases theV max of taurine uptake by brush border membrane vesicles. Pediatr Nephrol 1:9–15
Chesney RW, Lippincott S, Gusowski N, Padilla M, Zelikovic I (1986) Studies on renal adaptation to altered dietary amino acid intake: tissue taurine responses in nursing and adult rats. J Nutr 116: 1965–1976
Booth AG, Kenny AJ (1974) A rapid method for the preparation of microvilli from rabbit kidney. Biochem J 142:575–581
Glossman H, Neville DM Jr (1972) γ-Glutamyl transferase in kidney brush border membranes. FEBS Lett 19:340–344
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–253
Chesney R, Sacktor B, Rowen RR (1973) The binding ofd-glucose to the isolated rabbit renal brush border. J Biol Chem 218:2182–2191
Zelikovic I, Stejskal-Lorenz E, Lohstroh P, Budreau A, Chesney RW (1989) Anion dependence of taurine transport by rat renal brush-border membrane vesicles. Am J Physiol 256:F646-F655
Daniel WD (1983) Biostatistics: a foundation for analysis in the health sciences. Wiley, New York, pp 177–180
Zelikovic I, Chesney RW (1989) Sodium-coupled amino acid transport in renal tubule. Kidney Int 6:351–359
Wedeen RP, Weiner B (1973) The distribution ofp-aminohippuric acid in rat kidney slices. I. Tubular localization. Kidney Int 3: 205–213
Rozen R, Scriver CR, Mohyuddin F (1983) Hypertaurinuria in the C57BL/6J mouse: altered transport at the renal basolateral membrane. Am J Physiol 244:F150-F155
Mandla S, Scriver CR, Tenenhouse HS (1988) Decreased transport in renal basolateral membrane vesicles from hypertaurinuric mice. Am J Physiol 255:F88-F95
Chesney RW, Jax DK (1979) Developmental aspects of renal β-amino acid transport. II. Ontogeny of uptake and efflux processes and effect of anoxia. Pediatr Res 13:861–867
Olea RS, Pasantes-Morales H, Lázaro A, Cereijido M (1991) Osmolarity-sensitive release of free amino acids from cultured kidney cells (MDCK). J Membr Biol 121:1–9
Chesney RW, Scriver CR, Mohyuddin F, (1976) Localization of the membrane defect in transepithelial transport of taurine by parallel studies in vivo and in vitro in hypertaurinuric mice. J Clin Invest 57: 183–193
Uchida S, Nakanishi T, Moo Kwon H, Preston AS, Handler JS (1991) Taurine behaves as an osmolyte in Madin-Darby kidney cells Protection by polarized, regulated transport of taurine. J Clin Invest 88:656–662
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chesney, R.W., Budreau, A.M. Efflux of taurine from renal brush border membrane vesicles: Is it adaptively regulated?. Pediatr Nephrol 7, 35–40 (1993). https://doi.org/10.1007/BF00861561
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00861561