Abstract
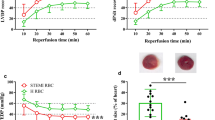
The aim of this study was to investigate if levels of circulating cyclic guanosine monophosphate (c-GMP) alter, in preconditioning. Twenty-eight rabbits were divided into four groups. In vivo hearts were preconditioned, either with 5 min (group A, n=8) or with 1 min (group B, n=8) ischemia, followed by 10 min reperfusion, while groups C (n=7) and D (n=5) had no interventions. Protection was determined by subjecting groups A, B and C (but not D) to 30 min regional ischemia which was followed (including group D) by 2h reperfusion. Seven blood samples were collected for the assessment of circulating c-GMP at different points of time. All results were expressed in pmol/ml using radioimmunoassay and the infarcted to risk area in percent using fluorescent particles and tetrazolium chloride (TTC). Circulating c-GMP increased during long ischemia only in group A (baseline value 47±4, long ischemic values 60.5±4 and 60.4±4, p<0.05). Circulating c-GMP in group A was significantly higher in the middle of the long ischemia in comparison to the groups B, C and D (60.5±4 vs 43.9±4, 45.8±5 and 43.6±4, p<0.05). Infarcted to risk ratio was lower in group A than in groups B and C (12.2±4 vs 29.6±6 and 34.2±6, respectively, p<0.05). Circulating c-GMP is increased in classically preconditioned in comparison to ineffectively preconditioned hearts or to control groups. This elevation may be related to the protective effect of this phenomenon.
Similar content being viewed by others
References
Ahlner J, Andersson RGG, Axelsson KL, Bergdahl B, Dahlstrom U, Swahn E (1988) Decreased plasma levels of cyclic-GMP in patients with chest pain and angiographically normal coronary arteries. Klin Wochenschr, 66: 494–497
Ashman DF, Lipton R, Milicow M, Price T (1963) Isolation of a denosine 3,5 monophosphate and guanosine 3,5 monophosphate from rat urine. Biochem Biophys Res Commun 11: 330–334
Casnellie JE (1991) Protein kinase inhibitors: probes for the functions of protein phosphorylation. Adv Pharmacol 22: 167–205
Davies PF, Tripathi SC (1993) Mechanical stress mechanisms and the cell. An endothelial paradigm. Circ Res 72: 239–245
Furchgott RF, Vanhoutte PM (1989) Endothelium-derived relaxing and contracting factors. The FASEB Journal 3: 2007–2017
Henderson AH (1991) Endothelium in control. Br H J 65: 116–125
Iliodromitis EK, Kremastinos DT, Apostolou T, Michelakakis N, Markianos M, Karavolias G, Kyriakides ZS, Toutouzas P (1992) Local coronary production of the endogenous vasodilator c-GMP during angioplasty. Eur Heart J 13S: 84
Iwamoto T, Miura T, Adachi T, Noto T, Ogawa T, Tsuchida, A, Lmmure O (1991) Myocardial infarct size-limiting effect of ischemic preconditioning was not attenuated by oxygen free radical scavengers in the rabbit. Circulation 83: 1015–1022
Jakob G, Mair J, Pichler M, Puschendorf B (1995) Ergometric, exercise testing and sensitivity of cyclic guanosine 3′, 5′-monophosphate (cGMP) in diagnosing asymptomatic left ventricular dysfunction. Br Heart J 73:145–150
Kondo T, Ogawa K, Ban M, Ogasawara B, Watanabe E, Satake T (1981) Plasma level of norepinephrine and cyclic nucleotides following acute myocardial infarction. Jpn Heart J 22: 593–603
Kyriakides ZS, Markianos M, Iliodromitis EK, Kremastinos DTh (1995) Vein plasma endothelin and cyclic-GMP increase during coronary angioplasty is related to myocardial ischemia. Eur Heart J 16: 894–898
Lawson CS, Downey JM (1993) Preconditioning; state of the art myocardial protection. Cardiovasc Res 27: 542–550
Li GC, Vasquez JA, Gallagher KP, Lucchesi BR (1990) Myocardial protection with preconditioning. Circulation 82: 609–619
Liu Y, Ytrehus K, Downey JM (1994) Evidence that translocation of protein kinase C is a key event during ischemic preconditioning of rabbit myocardium. J Mol Cell Cardiol 26: 661–668
Liu Y, Tsuchida A, Cohen MV, Downey JM (1995) Pretreatment with angiotensin II activates protein kinase C and limits myocardial infarction in isolated rabbit hearts. J Mol Cell Cardiol 27: 883–892
Malatino LS, Leonardi C, Stancanelli B, Polizzi G, Grassi R, Tamburino C, Tamburino G (1992) Transient myocardial ischemia stimulates atrial natriuretic factor release. Am Heart J 123: 693–698
Murad F (1986) Cyclic guanosine monophosphate as a mediator of vasodilation. J Clin Invest 78: 1–5
Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 74: 1124–1136
Parratt JR (1994) Protections of the heart by ischemic preconditioning: mechanisms and possibilities for pharmacological exploitation. TIPS 15: 19–25
Parratt JR (1995) Possibilities for the pharmacological exploitation of ischaemic preconditioning. J Mol Cell Cardiol 27: 991–1000
Patel VC, Woolfson RG, Singh KJ, Neild GH, Yellon DM (1992) Ischaemic preconditioning is not prevented by inhibition of endothelium-derived nitric oxide. J Mol Cell Cardiol 24 (I): 152
Radomski, MW, Palmer RMJ, Moncada S (1987) The role of nitric oxide and cGMP in platelet adhesion to vascular endothelium. Biochem Biophys Res Commun 148: 1482–1489
Sax FL, Cannon RO, Hanson C, Epstein SE (1987) Impaired forearm vasodilator reserve in patients with microvascular angina. NEJM 317: 1366–1370
Schmidt K, Mayer JB, Kukovetz WR (1989) Effect of calcium on endothelium-derived relaxing factor formation and c-GMP levels in endothelial cells. Eur J Pharmacol 170: 157–166
Schott RJ, Rohmann S, Braun ER, Schaper W (1990) Ischemic preconditioning reduces, infarct size in swine myocardium. Circ Res 66: 1133–1142
Schulz S, Chinkers M, Garbers DL (1989) The guanylate cyclase/receptor family of proteins. The FASEB Journal 3: 2026–2035
Stamler JS, Loscalzo J (1991) The antiplatelet effect of organic nitrates and related nitroso compounds and their relevance to cardiovascular disorders. JACC 18: 1529–1536
Szilvassy Z, Ferdinandy P, Bor P, Jakab I, Lonovics J, Koltai M (1994) Ventricular overdrive pacing-induced, anti-ischemic effect: a conscious rabbit model of preconditioning. Am J Physiol, 266 (35): H2033-H2041
Van Winkle DM, Thornton J, Downey JM (1991) Cardioprotection from ischemic preconditioning is lost following prolonged reperfusion in the rabbit. Coronary Artery Dis 2: 613–619
Vegh A, Szekeres L, Parratt J (1992) Preconditioning of the ischemic myocardium; involvement of the L-arginine nitric oxide pathway. Br J Pharmacol 107: 648–652
Woolfson RG, Patel VC, Neild G, Yellon DM (1995) Inhibition of nitric oxide synthesis reduces infarct size by an adenosine-dependent mechanism. Circulation 91: 1545–1551
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Iliodromitis, E.K., Papadopoulos, C.C., Markianos, M. et al. Alterations in circulating cyclic guanosine monophosphate (c-GMP) during short and long ischemia in preconditioning. Basic Res Cardiol 91, 234–239 (1996). https://doi.org/10.1007/BF00788909
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00788909