Abstract
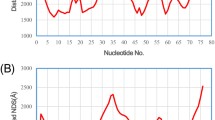
It is proposed that a general function of noncoding DNA and RNA sequences in higher organisms (intergenic and intervening sequences) is to provide multiple binding sites over long stretches of polynucleotide for certain types of regulatory proteins. Through the building up or abolishing of high-order structures, these proteins either sequester sites for the control of, e.g., transcription or make the sites available to local molecular signals. If this is to take place, the existence of a ‘c-value paradox’ becomes a requirement. Multiple binding sites for a given protein may recur in the form of a sequence ‘motif’ that is variable within certain limits. Noncoding sequences of the chicken ovalbumin gene furnish an appropriate example of a sequence motif, GAAAATT. Its improbably high frequency and significant periodicity are both absent from the coding sequences of the same gene and from the noncoding sequences of a differently controlled gene in the same organism, the preproinsulin gene. This distribution of a sequence motif is in keeping with the concepts outlined. Low specificity of sequences that bind protein is likely to be compatible with highly specific conformational changes.
Similar content being viewed by others
References
AmentaP. S., GershI. & GershE. S., 1973. In: I.Gersh (ed.) Submicroscopic Cytochemistry I. Academic Press, New York, pp. 365–375.
Artavanis-TsakonasS., SchedlP., MiraultM. E., MoranL. & LisJ., 1979. Cell 17: 9–18.
BakA. L. & ZeuthenJ., 1978. Cold Spring Harbor Symp. Quant. Biol. 1977. 42: 367–378.
BarnettT. & RaeP. M. M., 1979. Cell 16: 763–775.
BeermannW., 1972. In: W.Beermann (ed.) Developmental Studies on Giant Chromosomes. Springer, New York, pp. 1–33.
Benyajati, C. & Worcel, A. Cell 9: 393–407.
BlakeC. C. F., 1979. Nature 277: 598.
BramS., 1975. Biochimie 57: 1301–1306.
BramS., BaudyP., Butler-BrowneG. & IbelK., 1974. Biochimie 56: 1339–1341.
BridgesC. B., 1938. J. Hered. 29: 11–13.
BrittenR. J. & DavidsonE. H., 1976. Fed. Proc. 35: 2151–2157.
CalameK., RogersJ., EarlyP., DavisM., LivantD., WallR. & HoodL., 1980. Nature 284: 452–455.
CatinoJ. J., BuschH., DaskalY. & YeomanL. C., 1979. J. Cell Biol. 83: 462–467.
CrickF., 1979. Science 204: 264–271.
DavidsonE. H. & BrittenR. J., 1979. Science 204: 1052–1059.
DickersonR. E., 1971. J. Mol. Evol. 1: 26–45.
DicksonE., BoydJ. B. & LairdC. D., 1971. J. Mol. Biol. 61: 615–628.
DoolittleW. F. & SapienzaC., 1980. Nature 284: 601–603.
Eastman, E. M., Goodman, R. M., Erlanger, B. F. & Miller, O. J., 1977. J. Cell Biol. 27: 149a.
EatonW. A., 1980. Nature 284: 183–185.
EdenF. C. & HendrickJ. P., 1978. Biochemistry 17: 5838–5844.
ElginS. C. R., SerunianL. A. & SilverL. M., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 839–849.
FinchJ. T. & KlugA., 1976. Proc. Natl. Acad. Sci. 73: 1897–1901.
FritschE. F., LawnR. M. & ManiatisT., 1980. Cell 19: 959–972.
FyrbergE. A., KindleK. L., DavidsonN. & SodjaA., 1980. Cell 19: 365–378.
GarelA. & AxelR., 1976. Proc. Natl. Acad. Sci. 73: 3966–3970.
GershI., 1973. In: I.Gersh (ed.) Submicroscopic Cytochemistry I. Academic Press, New York, pp. 221–282.
GershE. S. & GershI., 1973. In: I.Gersh (ed.) Submicroscopic Cytochemistry I. Academic Press, New York, pp. 163–194.
GilbertW., 1978. Nature 271: 501.
GloverD. M. & HognessD. S., 1977. Cell 10: 167–176.
GoughN. M., KempD. J., TylerB. M., AdamsJ. M. & CoryS., 1980. Proc. Natl. Acad. Sci. 77: 554–558.
GrussP. & KhouryG., 1980. Nature 286: 634–637.
HamerD. H. & LederP., 1979. Cell 18: 1299–1302.
Igo-KemenesT. & ZachauH. G., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 109–118.
JacqC., LazowskaJ. & SlonimskiP. P., 1980. Compt. Rend. Acad. Sci., D, 290: 89–92.
KeppelF., AlletB. & EisenH., 1977. Proc. Natl. Acad. Sci. 74: 653–656.
KroegerH. & MüllerG., 1973. Exp. Cell Res. 82: 89–94.
LauerJ., ShenC. M. J. & ManiatisT., 1980. Cell 20: 119–130.
LevyW. B., WongN. C. W. & DixonG. H., 1977. Proc. Natl. Acad. Sci. 74: 2810–2814.
LezziM. & RobertM., 1972. Res. Probl. Cell Diff. 4: 35–57.
LomedicoP., RosenthalN., EfstratiadisA., GilbertW., KolodnerR., & TizardR., 1979. Cell 18: 545–558.
MarsdenM. P. F. & LaemmliU. K., 1979. Cell 17: 849–858.
MaurizotJ. C., CharlierM. & HeleneC., 1974. Biochem. Biophys. Res. Comm. 60: 951–957.
MayfieldJ. E., SerunianL. A., SilverL. M. & ElginS. C. R., 1978. Cell 14: 539–544.
MiyataT., YasunagaT., Yamawaki-KataokaY., ObataM. & HonjoT., 1980. Proc. Natl. Acad. Sci. 77: 2143–2147.
MusichP. R., BrownF. L. & MaioJ. J., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 1147–1160.
NunbergJ. H., KaufmanR. J., ChangA. C. Y., CohenS. N. & SchimkeR. T., 1980. Cell 19: 355–364.
OrgelL. E. & CrickF., 1980. Nature 284: 604–607.
PalmiterR. D., MulvihillE. R., McKnightG. S., & SenearA. W., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’ 42: 639–647.
PaulsonJ. R. & LaemmliU. K., 1977. Cell 12: 817–828.
PerlerF., EfstratiadisA., LomedicoP., GilbertW., KolodnerR. & DodgsonJ., 1980. Cell 20: 555–566.
PrunellA. & KornbergR. D., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 103–108.
RenzM., NehlsP. & HozierJ., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 245–252.
RobertsonM. A., StadenR., TanakaY., CatterallJ. F., O'MalleyB. W., & BrownleeG. G., 1979. Nature 278: 370–372.
RosbashM., CampoM. S. & GummersonK. S., 1975. Nature 258: 682–686.
SakanoH., HuppiK., HeinrichG. & TonegawaS., 1979. Nature 280: 288–294.
Schibler, U. Communicated at Fourth Arolla Workshop, EMBO, August 1980.
SedatJ. & ManuelidisL., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 331–350.
SilverL. M. & ElginS. C. R., 1977. Cell 11: 971–983.
SlonimskiP., 1980. Compt. Rend. Acad. Sci. 290: 331–334.
SmithM., LeungD. W., GillamS., AstellC. R., MontgomeryD. L. & HallB. D., 1979. Cell 16: 753–762.
SorsaV., 1974. Cold Spring Harbor Symp. Quant. Biol. ‘1973’. 38: 601–608.
SorsaM. & SorsaV., 1967. Chromosoma 22: 32–41.
SorsaM. & SorsaV., 1970. Chromosoma 31: 346.
SperlingL. & KlugA., 1977. J. Mol. Biol. 112: 253–263.
StalderJ., GroudineM., DodgsonJ. B., EngelJ. D. & WeintraubH., 1980. Cell 19: 973–980.
SuauP., BradburyE. M. & BaldwinJ. P., 1979. Eur. J. Biochem. 97: 593–602.
TakahataN. & MaruyamaT., 1979. Proc. Natl. Acad. Sci. 76: 4521–4525.
ThomaF. & KollerT., 1977. Cell 12: 101–107.
TiemeierD. C., TilghmanS. M., PolskyF. I., SeidmanJ. G., LederA., EdgellM. H. & LederP., 1978. Cell 14: 237–245.
VanOoyenA., BergJ.van den, ManteiN. & WeissmanC., 1979. Science 206: 337–344.
WeintraubH. & GroudineM., 1976. Science 193: 848–856.
WeisbrodS., GroudineM. & WeintraubH., 1980. Cell 19: 289–301.
WeisbrodS. & WeintraubH., 1979. Proc. Natl. Acad. Sci. 76: 630–634.
WorcelA., 1978. Cold Spring Harbor Symp. Quant. Biol. ‘1977’. 42: 313–324.
WuC., 1980. Nature 286: 854–860.
YoungN. S., BenzE. J., KantorJ. A., KretschmerP. & NienhuisA. W., 1978. Proc. Natl. Acad. Sci. 75: 5884–5888.
ZuckerkandlE., 1976. J. Mol. Evol. 9: 73–104.
ZuckerkandlE., 1978. J. Mol. Evol. 12: 57–89.
ZuckerkandlE., 1979. J. Mol. Evol. 14: 311–321.
ZuckerkandlE. & PaulingL., 1962. In: M.Kasha and B.Pullman (eds.) Horizons in Biochemistry. Academic Press, New York, pp. 189–225.
ZuckerkandlE. & PaulingL., 1965. In: V.Bryson & H. J.Vogel (eds.), Evolving Genes and Proteins, Academic Press, New York, pp. 97–166.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zuckerkandl, E. A general function of noncoding polynucleotide sequences. Mol Biol Rep 7, 149–158 (1981). https://doi.org/10.1007/BF00778746
Issue Date:
DOI: https://doi.org/10.1007/BF00778746