Abstract
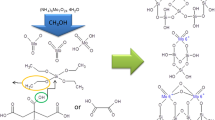
In contrast to the frequently reported lack of interaction between hexavalent molybdenum and SiO2 and the tendency of silica-supported MoO3 to coalescence, it has been found that on dehydration small molybdenum oxide clusters spread over a silica support. A combined Raman spectroscopy-X-ray absorption study shows a significantly altered structure of the molybdenum oxide phase after dehydration. In EXAFS the total Mo-Mo coordination number drops from 3.27 to 0.20 after anin situ thermal treatment at 673 K. The increase of the peak in the XANES region (Is -→ 4d) indicates that the coordination sphere of the molybdenum atoms strongly alters after dehydration. The Raman spectra reflect the change of the structure through a shift of the position of the terminal Mo=O bond from 944 to 986 cm−1 and the disappearance of the bridged Mo-O-Mo vibration at 880 cm−1. It is concluded that dehydration produces almost isolated molybdenum sites in this highly dispersed sample. Water ligands stabilize the oligomeric clusters under ambient conditions; the removal of water causes spreading of these clusters.
Similar content being viewed by others
References
N. Kakuta, K. Tohji and Y. Udagawa, J. Phys. Chem. 92 (1988) 2583.
S.R. Stampfl, Y. Chen, J.A. Dumesic, C. Niu and C.G. Hill, J. Catal. 105 (1987) 445.
G.A. Tsigdinos, H.Y. Chen and B.J. Streusand, Ind. Eng. Chem. Prod. Res. Dev. 20 (1981) 619.
Y. Barbaux, A.R. Elamrani, E. Payen, L. Gengembre, J.P. Bonnelle and B. Grzybowska, Appl. Catal. 44 (1988) 117.
M. Anpo, M. Kondo, Y. Kubokawa, C. Louis and M. Che, J. Chem. Soc., Faraday Trans, I, 84(8) (1988) 2771.
S.R. Seyedmonir, S. Abdo and R.F. Howe, J. Phys. Chem. 86 (1982) 1233.
J.M. Stencel, J.R. Diehl, J.R. d'Este, L.E. Makovsky, L. Rodrigo, K. Marcinkowska, A. Adnot, P.C. Roberge and S. Kaliaguine, J. Phys. Chem. 90 (1986) 4739.
J. Leyrer, M.I. Zaki and H. Knözinger, J. Phys. Chem. 90 (1986) 4775.
J. Leyrer, D. Mey and H. Knözinger, J. Catal. 124 (1990) 349.
T. Machej, J. Haber, A. Turek and I.E. Wachs, to be published.
L. Rodrigo, A. Adnot, P.C. Roberge and S. Kaliaguine, J. Catal. 105 (1987) 175.
H. Jeziorowski and H. Knözinger, J. Phys. Chem. 83(9) (1979) 1166.
J.A.R. van Veen and P.A.J.M. Hendriks, Polyhedron 5 (1986) 75.
C.T.J. Mensch, J.A.R. van Veen, B. van Wingerden and M.P. van Dijk, J. Phys. Chem. 92 (1988) 4961.
Y. Okamoto and T. Imanaka, J. Phys. Chem. 92 (1988) 7102.
H. Praliaud, J. Less Common Metals 54 (1977) 387.
P. Sarrazin, B. Mouchel and S. Kasztelan, J. Phys. Chem. 93 (1989) 904.
E.T.C. Vogt, A.J. van Dillen, J.W. Geus, J.J.P. Biermann and F.J.J.G. Janssen, in:Proc. 9th I.C.C., eds. M.J. Phillips and M. Ternan, Vol. 4, Calgary, 1988, p. 1976.
L. Wang and W.K. Hall, J. Catal. 82 (1983) 177.
F.W.H. Kampers, T.M.J. Maas, J. van Grondelle, P. Brinkgreve and D.C. Koningsberger, Rev. Sci. Instr. 60 (1989) 2635.
J.B.A.D. Van Zon, D.C. Koningsberger, H.F.J. Van 't Blik and D.E. Sayers, J. Chem. Phys. 82 (1985) 5742.
S.M.A. Bouwens, R. Prins, V.H.J. de Beer and D.C. Koningsberger, J. Phys. Chem. 94 (1990) 3711.
W.P. Griffith and P.J.B. Lesniak, J. Chem. Soc. (A) (1969) 1066.
G. Deo and I.E. Wachs, J. Phys. Chem. accepted.
T. Machej, J. Haber, A.M. Turek and I.E. Wachs, Appl. Catal. 70 (1991) 115.
N. Nakamoto,Infrared and Raman spectroscopy of Inorganic and Coordination Compounds (Wiley, New York, 1978).
F.D. Hardcastle and I.E. Wachs, J. Raman Spectrosc. 21 (1990) 683.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Boer, M., van Dillen, A.J., Koningsberger, D.C. et al. Remarkable spreading behavior of molybdena on silica catalysts. Anin situ EXAFS-Raman study. Catal Lett 11, 227–239 (1991). https://doi.org/10.1007/BF00764089
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00764089