Abstract
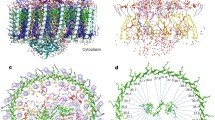
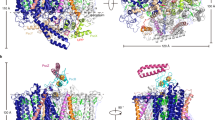
The three-dimensional structure of the photosynthetic reaction center fromRhodobacter sphaeroides is described. The reaction center is a transmembrane protein that converts light into chemical energy. The protein has three subunits: L, M, and H. The mostly helical L and M subunits provide the scaffolding and the finely tuned environment in which the chromophores carry out electron transfer. The details of the protein-chromophore interactions are from studies of a trigonal crystal form that diffracted to 2.65-Å resolution. Functional studies of the multi-subunit complex by site-specific replacement of key amino acid residues are summarized in the context of the molecular structure.
Similar content being viewed by others
References
Allen, J. P., Feher, G., Yeates, T. O., Rees, D. C., Deisenhofer, J., Michel, H., and Huber, R. (1986).Proc. Natl. Acad. Sci. USA 83 8589–8593.
Allen, J. P., Feher, G., Yeates, T. O., Komiya, H., and Rees, D. C. (1987)Proc. Natl. Acad. Sci. USA 84 6162–6166.
Belanger, G., Berard, J., Corriveau, P., and Gingras, G. (1988).J. Biol. Chem. 263 7632–7638.
Brünger, A. T., Kuriyan, J., and Karplus, M. (1987).Science 235 458–460.
Buchanan, S. K., Fritzsch, G., Ermler, U., and Michel, H. (1993).J. Mol. Biol. 230 1311–1314.
Bylina, E. J., Kirmaier, C., McDowell, L., Holten, D., and Youvan, D. C. (1988).Nature (London)336 182–184.
Bylina, E. J., and Youvan, D. C. (1988).Proc. Natl. Acad. Sci. USA 85 7226–7230.
Bylina, E. J., Kolaczkowski, S. V., Norris, J. R., and Youvan, D. C. (1990).Biochemistry 29 6203–6210.
Chan, C.-K., Chen, L. X.-Q., DiMagno, T. J., Hanson, D. K., Nance, S. L., Schiffer, M., Norris, J. R., and Fleming, G. R. (1991).Chem. Phys. Lett. 176 366–372.
Chang, C.-H., Schiffer, M., Tiede, D., Smith, U., and Norris, J. (1985).J. Mol. Biol. 186 201–203.
Chang, C.-H., Tiede, D., Tang, J., Smith, U., Norris, J., and Schiffer, M. (1986).FEBS Lett. 205 82–86.
Chang, C.-H., El-Kabbani, O., Tiede, D., Norris, J., and Schiffer, M. (1991).Biochemistry 30 5352–5360.
Chory, J. T., Donohue, J., Varga, A. R., Staehelin, L. A., and Kaplan, S. (1984).J. Bacteriol. 159 540–554.
Coleman, W. J., and Youvan, D. C. (1990).Annu. Rev. Biophys. Biophys. Chem. 19 333–367.
Debus, R. J., Feher, G., and Okamura, M. Y. (1985).Biochemistry 24 2488–2500.
Deisenhofer, J., Epp, O., Miki, R., Huber, R., and Michel, H. (1985),Nature (London)318 618–624.
Deisenhofer, J., and Michel, H. (1989).EMBO J. 8 2149–2169.
DiMagno, T. J., Rosenthal, S. J., Xie, X., Du, M., Chan, C.-K., Hanson, D. K., Schiffer, M., Norris, J. R., and Fleming, G. R. (1992). InThe Photosynthetic Bacterial Reaction Center II (Breton, J., and Vermeglio, A., eds.), Plenum Press, New York, pp. 209–217.
Ermler, U., Fritzsch, G., Buchanan, S., and Michel, H. (1992). InResearch in Photosynthesis (Murata, N., ed.), Kluwer, The Netherlands, pp. 341–347.
Farchaus, J. W., and Oesterhelt, D. (1989).EMBO J. 8 47–54.
Feher, G., Allen, J. P., Okamura, M. Y., and Rees, D. C. (1989).Nature (London)339 111–116.
Gray, K. A., Farchaus, J. W., Wachtveitl, J., Breton, J., and Oesterhelt, D. (1990).EMBO J. 9 2061–2070.
Hanson, D. K., Tiede, D. M., Nance, S. L., Chang, C.-H., and Schiffer, M. (1993).Proc. Natl. Acad. Sci. USA.,90 8929–8933.
Hendrickson, W. (1985).Methods Enzymol. 115 252–270.
Kraulis, P. J. (1991).J. Appl. Crystallogr. 24 946–950.
Laussermair, E., and Oesterhelt, D. (1992).EMBO J. 11 777–783.
Leibl, W., Sinning, I., Ewald, G., Michel, H., and Breton, J. (1993).Biochemistry 32 1958–1964.
Luzzati, P. V. (1952).Acta Crystallogr. 5 802–810.
McDowell, L. M., Gaul, D., Kirmaier, C., Holten, D., and Schenck, C. C. (1991).Biochemistry 30 8315–8322.
Michel, H., Weyer, K. A., Gruenberg, H., and Lottspeich, F. (1985).EMBO J. 4 1667–1672.
Michel, H., Weyer, K. A., Gruenberg, H., Dunger, I., Oesterhelt, D., and Lottspeich, F. (1986a).EMBO J. 5 1149–1158.
Michel, H., Epp, O., and Deisenhofer, J. (1986b).EMBO J. 5 2445–2451.
Michel, H., Sinning, I., Koepke, J., Ewald, G., and Fritzsch, G. (1990).Biochim. Biophys. Acta 1018 115–118.
Michel-Beyerle, M. E., Plato, M., Deisenhofer, J., Michel, H., Bixon, M., and Jortner, J. (1988).Biochim. Biophys. Acta 932 52–70.
Murchison, H. A., Alden, R. G., Allen, J. P., Peloquin, J. M., Taguchi, A. K. W., Woodbury, N. W., and Williams, J. C. (1993).Biochemistry 32 3498–3505.
Nagarajan, V., Parson, W. W., Gaul, D., and Schenck, C. (1990).Proc. Natl. Acad. Sci. USA 87 7888–7892.
Okamura, M. Y., and Feher, G. (1992).Annu. Rev. Biochem. 61 861–896.
Ovchinnikov, Y. A., Abdulaev, N. G., Zolotarev, A. S., Schmuckler, B. E., Zargarov, A. A., Kutuzov, M. A., Telezhinskaya, I. N., and Levina, N. B. (1988a).FEBS Lett. 231 237–242.
Ovchinnikov, Y. A., Abdulaev, N. G., Shmuckler, B. E., Zargarov, A. A., Kutuzov, M. A., Telezhinskaya, I. N., Levina, N. B., and Zolotarev, A. S. (1988b).FEBS Lett. 232 364–368.
Paddock, M. L., Rongey, S. H., Feher, G., and Okamura, M. Y. (1989).Proc. Natl. Acad. Sci. USA 86 6602–6606.
Paddock, M. L., McPherson, P. H., Feher, G., and Okamura, M. Y. (1990).Proc. Natl. Acad. Sci. USA 87 6803–6807.
Read, R. J. (1986).Acta Crystallogr. Sect. A 42, 140–149.
Schiffer, M., Chang, C.-H., and Stevens, F. J. (1992a).Protein Eng. 5 213–214.
Schiffer, M., Chan, C.-K., Chang, C.-H., DiMagno, T. J., Fleming, G. R., Nance, S., Norris, J., Snyder, S., Thurnauer, M., Tiede, D. M., and Hanson, D. K. (1992b). InThe Photosynthetic Bacterial Reaction Center II (Breton, J., and Vermeglio, A., eds.), Plenum Press, New York, pp. 351–361.
Shiozawa, J. A., Feick, R., Oesterhelt, D., and Lottspeich, F. (1989).Eur. J. Biochem 180 75–84.
Stilz, H. U., Finkele, U., Holzapfel, W., Lauterwasser, C., Zinth, W., and Oesterhelt, D. (1993).Eur. J. Biochem., submitted.
Takahashi, E., Maroti, P., and Wraight, C. A. (1990). InCurrent Research in Photosynthesis (Baltscheffsky, M., ed.), Kluwer, The Netherlands, pp. 169–172.
Takahashi, E., and Wraight, C. A. (1992).Biochemistry 31 855–866.
Tiede, D. M., Budil, D. E., Tang, J., El-Kabbani, O., Norris, J. R., Chang, C.-H., and Schiffer, M. (1988). InThe Photosynthetic Bacterial Reaction Center (Breton, J., and Vermeglio, A., eds.), Plenum Press, New York, pp. 13–20.
Tronrud, D. E., Ten Eyck, L. F., and Matthews, B. W. (1987).Acta Crystallogr. Sect. A 43, 489–501.
Weyer, K. A., Lottspeich, F., Gruenberg, H., Lang, F., Oesterhelt, D., and Michel, H. (1987).EMBO J. 6 2197–2202.
Williams, J. C., Steiner, L. A., Ogden, R. C., Simon, M. I., and Feher, G. (1983).Proc. Natl. Acad. Sci. USA 80 6505–6509.
Williams, J. C., Steiner, L. A., Feher, G., and Simon, M. I. (1984).Proc. Natl. Acad. Sci. USA 81 7303–7307.
Williams, J. C., Steiner, L. A., and Feher, G. (1986).Proteins 1 312–325.
Yeates, T. O., Komiya, H., Chirino, A., Rees, D. C., Allen, J. P., and Feher, G. (1988).Proc. Natl. Acad. Sci. USA 85 7993–7997.
Youvan, D. C., Bylina, E. J., Alberti, M. Begusch, H., and Hearst, J. E. (1984).Cell 37 949–957.
Youvan, D. C., Ismail, S., and Bylina, E. J. (1985).Gene 33 19–30.
Author information
Authors and Affiliations
Additional information
This work was supported in part by the U.S. Department of Energy, Office of Health and Environmental Research, under Contract No. W-31-109-ENG-38 and by Public Health Service Grant GM36598.
Rights and permissions
About this article
Cite this article
Ermler, U., Michel, H. & Schiffer, M. Structure and function of the photosynthetic reaction center fromRhodobacter sphaeroides . J Bioenerg Biomembr 26, 5–15 (1994). https://doi.org/10.1007/BF00763216
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00763216