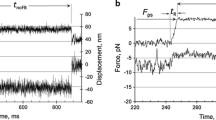
Summary
The change in orientation of myosin crossbridges in contracting muscle during sudden length changes was examined by fluorescence polarization. This study used a fluorescent ATP analogue, 1,N 6-etheno-2-aza-ATP(ε-2-aza-ATP) as a probe. Its fluorescence is considerably enhanced upon binding with myosin and is dependent on the chemical state of the myosin-nucleotide complex in muscle. The results showed that nucleotides bound to crossbridges in the intermediate attached state (presumably AM-ε-2-aza-ADP-Pi) during isometric contraction are highly oriented at the same angle as that of AM in rigor with bound ε-2-aza-ADP. Furthermore the orientation of nucleotides bound to crossbridges in the attached state is not altered during sudden changes in length of isometrically contracting muscle. The results of this time-resolved measurement support the conclusion obtained from a previous steady-state experiment that change in axial orientation of the active site of the myosin head is not involved in force generation.
Similar content being viewed by others
References
ARATA, T., MUKOHATA, Y. & TONOMURA, Y. (1977) Structure and function of the two heads of the myosin molecule VI. ATP hydrolysis, shortening and tension development of myofibrils.J. Biochem. 82, 801–12.
ARATA, T. & SHIMIZU, H. (1980) Spin-label study of actin-myosin-nucleotide interactions glycerinated muscle fibers.J. molec. Biol. 151, 411–37.
COOKE, R., CROWDER, M. S. & THOMAS, D. D. (1982) Orientation of spin-labels attached to crossbridges in contracting muscle fiber.Nature 300, 776–8.
HUXLEY, A. F. (1974) Review lecture on muscle contraction.J. Physiol. 243, 1–43.
HUXLEY, A. F. & SIMMONS, R. M. (1971) Proposed mechanism of force generation in striated muscle.Nature 233, 533–8.
HUXLEY, H. E., FARUQI, A. R., KRESS, M., BORDAS, J. & KOCH, M. H. J. (1982) Time-resolved X-ray diffraction studies of the myosin layer-line reflections during muscle contraction.J. molec. Biol. 158, 637–84.
HUXLEY, H. E., SIMMONS, R. M., FARUQI, A. R., KRESS, M., BORDAS, J. & KOCH, M. H. J. (1981)Proc. natn. Acad. Sci. U.S.A. 78, 2297–301.
HUXLEY, H. E., SIMMONS, R. M., FARUQI, A. R., KRESS, M., BORDAS, J. & KOCH, M. H. J. (1983) Changes in the X-ray reflections from contracting muscle during rapid mechanical transients and their structural implications.J. molec. Biol. 169, 469–506.
MARSTON, S. B. & TREGEAR, R. T. (1972) Evidence for a complex between myosin and ADP in relaxed muscle fibers.Nature 235, 23–5.
MIYATA, H. & ASAI, H. (1982) 1,N 6-etheno-2-aza-adenosine triphosphate: Enormous increase in fluorescence intensity induced by its binding to heavy meromyosin and estimation of the kinetic parameters.Biochem. biophys. Res. Commun. 105, 269–302.
NAGANO, H. & YANAGIDA, T. (1984) Predominant attached state of myosin cross-bridges during contraction and relaxation at low ionic strength.J. molec. Biol. 177, 769–85.
PRÓCHNIEWICZ-NAKAYAMA, E. & YANAGIDA, T. (1984) Oriented polymerization of actin in muscle ghost fibre. InAbstracts of the 22nd Japan Biophysical Conference, p. 577.
THOMAS, D. D. & COOKE, R. (1980) Orientation of spin-labeled myosin heads in glycerinated muscle fibers.Biophys. J. 32, 891–906.
TREGEAR, R. T. & SQUIRE, J. M. (1973) Myosin content and filament structure in smooth and striated muscle.J. molec. Biol. 77, 279–90.
TUSO, K. C., MILLER, E. E. & LO, K. W. (1974) Synthesis of 1,N 6-etheno-2-aza-adenosine(2-aza-ε-adenosine): a new cytotaxic fluorescent nucleotide.Nucleic Acid Res. 1, 531–47.
WILSON, M. G. A. & MENDELSON, R. A. (1983) A comparison of order and orientation of crossbridges in rigor and relaxed muscle fibers using fluorescence polarization.J. Musc. Res. Cell Motility 4, 671–93.
YANAGIDA, T. (1980) The angles of cross-bridges bound to nucleotide at various concentrations of Mg-(ε)-ATP. InMuscle Contraction: Its Regulatory Mechanisms (edited by EBASHI, S., MARUYAMA, K. and ENDO, M.), pp. 173–80. Tokyo: Japanese Scientific Press/Berlin: Springer-Verlag.
YANAGIDA, T. (1981) Angles of nucleotides bound to cross-bridges in glycerinated muscle fiber at various concentrations of ε-ATP, ε-ADP and ε-AMPPNP detected by polarized fluorescence.J. molec. Biol. 146, 539–60.
YANAGIDA, T. (1984a) Neo-sliding mechanism of muscle contraction and cell motility. InAbstracts of the 10th Yamada International Conference on Cell Motility II: Mechanism and Regulation (edited by ISHIKAWA, H.). Tokyo: University of Tokyo Press. (In press).
YANAGIDA, T. (1984b) Angles of fluorescently labelled myosin heads and actin monomers in contracting and rigor stained muscle fiber. InCross-bridge Mechanisms in Muscle Contraction (edited by POLLACK, G. H. and SUGI, H.), pp. 397–411. New York: Plenum Press.
YANAGIDA, T., KURANAGA, I. & INOUE, A. (1982) Interaction of myosin with thin filaments during contraction and relaxation: effect of ionic strength.J. Biochem. 92, 407–12.
YANAGIDA T. & OOSAWA, F. (1978) Polarized fluorescence from ε-ADP incorporated into F-actin in a myosin-free single fiber: conformation of F-actin and changes induced in it by heavy meromyosin.J. molec. Biol. 126, 507–24.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yanagida, T. Angle of active site of myosin heads in contracting muscle during sudden length changes. J Muscle Res Cell Motil 6, 43–52 (1985). https://doi.org/10.1007/BF00712310
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00712310