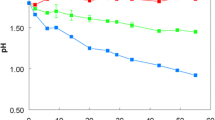
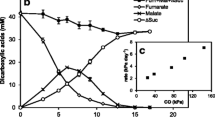
Summary
Fluoroacetate was extremely toxic toThiobacillus neapolitanus strainC, retarding growth even at 10-9 m. Inhibition could be relieved by relatively high concentrations of acetate or propionate. Inhibited cultures eventually recovered from inhibition and grew in the presence of fluoroacetate over the concentration range 10-9 to 10-5 m. The recovery represented a recommencement of division of the total population, as it was shown that 60–100% of the organisms inoculated on to agar containing as much as 10-3 m fluoroacetate formed colonies after lags as long as 37 days. Even longer lags occurred with more fluoroacetate, but fewer organisms survived. Fluoroacetate appeared specifically to inhibit the Krebs' cycle through fluorocitrate synthesis; this confirmed that the cycle is essential to the autotrophic metabolism.
Fluoroacetate-resistant variants occurred spontaneously at a frequency of about 2 per million organisms. These grew at normal exponential rates even in the presence of 10-2 m fluoroacetate. They appeared to differ from the wild type organism only in lacking acetyl coenzyme A synthetase and possibly having decreased permeability to acetate. The origin of acetyl coenzyme A for biosynthesis in these mutants, and the significance of the lack of “heterotrophic enzymes” from an obligate autotroph, are discussed.
Similar content being viewed by others
References
Aldous, J. G.: The nature of the metabolites of fluoroacetic acid in Baker's Yeast. Biochem. Pharmacol.12, 627–632 (1963).
—, and K. R.Rozee: The effect of pH on the toxicity of fluoroacetic acid. Biochem. J.62, 605–610 (1956).
Elliott, W. B., and G.Kalnitsky: A mechanism for fluoroacetate inhibition. J. biol. Chem.186, 487–493 (1950).
Fieser, L. F., and M.Fieser: Organic Chemistry, p. 163. New York: Reinhold Publishing Corporation 1956.
Ghunau, J. A., and I. C.Gunsalus: Conversion of pyruvate to acetylcoenzyme A. Biochemists' Handbook (Ed. C.Long), pp. 547–551. London: E. & F. N. Spon Ltd. 1961.
Hoare, D. S.: The breakdown and biosynthesis of glutamic acid. J. gen. Microbiol.32, 157–161 (1963).
Kalnitsky, G., and E. S. G.Barron: The effect of fluoroacetate on the metabolism of yeast and bacteria. J. biol. Chem.170, 83–95 (1947).
Kelly, M.: Isolation of bacteria able to metabolize fluoroacetate or fluoroacetamide. Nature (Lond.)208, 809–810 (1965).
Kelly, D. P.: Assimilation of organic compounds by a strictly chemoautotrophicThiobacillus. J. gen. Microbiol.41V (1965).
Kelly, D. P.: Problems of the autotrophic microorganisms. Sci. Progr.55, 35–51 (1967a).
Kelly, D. P.: Influence of amino acids and organic antimetabolites on growth and biosynthesis of the chemoatotrophThiobacillus neapolitanus strainC. Arch. Mikrobiol.56, 91–105 (1967b).
—: The incorporation of acetate by the chemoautotrophThiobacillus neapolitanus strainC. Arch. Mikrobiol.58, 99–116 (1967c).
—, and P. J.Syrett: The effect of uncoupling agents on carbon dioxide fixation by aThiobacillus. J. gen. Microbiol.34, 307–317 (1964).
Lotspeich, W. D., R. A. Peters, and T. W.Wilson: Inhibition of aconitase by the “active” factor formed from fluoroacetate in poisoned kidney tissue. J. Physiol. (Lond.)115, 24P (1951).
Lowry, O. H., N. J.Rosebrough, A. L.Farr, and R. J.Randall: Protein measurement with the Folin phenol reagent. J. biol. Chem.193, 265–275 (1951).
Mager, J., J.Goldblum-Sinai, and I.Blank: Effect of fluoracetic acid and allied fluoroanalogues on growth ofEscherichia coli. J. Bact.70, 320–325 (1955).
Marcus, A., and W. B.Elliott: Enzymatic reactions of fluoroacetate and fluoroacetyl coenzyme A. J. biol. Chem.218, 823–830 (1956).
——: Enzymatic reactions of fluoroacetyl phosphate. J. biol. Chem.234, 1011–1014 (1959).
Merrett, M. J., and P. J.Syrett: The relationship between glucose oxidation and acetate oxidation inChlorella vulgaris. Physiol. Plantarum (Copenh.)13, 237–249 (1960).
Morrison, J. F., and R. A.Peters: Biochemistry of fluoroacetate poisoning: the effect of fluorocitrate on purified aconitase. Biochem. J.58, 473–479 (1954).
Peters, R. A.: Lethal synthesis. Proc. roy. Soc. B139, 143–170 (1951).
—: Mechanism of the toxicity of the active constituent ofDichopetalum cymosum and related compounds. Advanc. Enzymol.18, 113–159 (1957).
—, R. W.Wakelin, and P.Buffa: Biochemistry of fluoroacetate poisoning. The isolation and some properties of the fluorotricarboxylic acid inhibitor of citrate metabolism. Proc. roy. Soc. B140, 497–507 (1953).
Smith, A. J., and J.Lascelles: Thiosulphate metabolism and rhodanese inChromatium sp. strainC. J. gen. Microbiol.42, 357–370 (1966).
Smith, I.: Chromatographic and electrophoretic techniques, Vol. 1. London: W. Heinemann 1960.
Stadtman, E. R.: The net synthesis of acetyl coenzyme A. J. biol. Chem.196, 535–546 (1952).
Still, G. G.: The role of some of the Krebs cycle reactions in the biosynthetic functions ofThiobacillus thioparus. Ph. D. thesis: Oregon State University (1965).
Tranter, E.Karen, and R. B.Cain: The degradation of fluoroaromatic compounds to fluorociatrate and fluoroacetate by bacteria. Biochem. J.103, 22P-23P (1967).
Trudinger, P. A.: Products of the anaerobic metabolism of tetrathionate byThiobacillus X. Aust. J. biol. Sci.17, 446–458 (1964).
—: Thiosulphate oxidation and cytochromes inThiobacillus X. I and II. Biochem. J.78, 673–680, 680–686 (1961).
van Niel, C. B.: The chemoautotrophic and photosynthetic sulphur bacteria. Ann. Rev. Microbiol.8, 105–132 (1954).
Wolleman, M., and G.Feuer: Formation of fluoroacetyl coenzyme A and fluoroacetylcholine from fluoroacetic acid and fluorocitric acid in brain extracts. Acta physiol. Acad. Sci. hung.11, 165–172 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kelly, D.P. Fluoroacetate toxicity inThiobacillus neapolitanus and its relevance to the problem of obligate chemoautotrophy. Archiv. Mikrobiol. 61, 59–76 (1968). https://doi.org/10.1007/BF00704292
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00704292