Summary
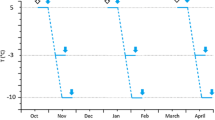
Overwintering larvae and adults of the stag beetle,Ceruchus piceus, are freeze sensitive (i.e. cannot survive internal freezing). The most commonly described cold adaptation of freeze susceptible insects involves the production of antifreezes to promote supercooling, butCeruchus piceus larvae produced only low levels of antifreezes in the winter. However, by removing ice nucleators from the gut and hemolymph in the winter the larvae were able to depress their supercooling points from approximately −7°C in the summer to near −25°C in mid-winter. The ice nucleators present in the non-winter hemolymph were identified as lipoproteins. One of these lipoproteins with ice nucleator activity was purified using flotation ultracentrifugation and anion exchange (DEAE-Sephadex) chromatography.
Removal of ice nucleators to promote supercooling in winter may be energetically preferable to costly production and maintenance of high, of-ten molar, concentrations of antifreeze. Obviously the ice nucleator must normally perform a function which the insect can spare over the winter. Hemolymph lipoproteins, which generally function in lipid transport, may fit this criterion during the winter period of reduced metabolic activity.
Similar content being viewed by others
Abbreviations
- LP I :
-
very low density lipoprotein
- LP II :
-
low density lipoprotein
- PAGE :
-
polyacrylamide gel electrophoresis
- SCP :
-
supercooling point
References
Bakken H (1985) Cold hardiness in the alpine beetlesPatrobus septentrionis andCalathus melanocephalus. J Insect Physiol 31:447–453
Baust JG (1976) Temperature buffering in an arctic microhabitat. Ann Ent Soc Am 69:117–119
Baust JG, Zachariassen KE (1983) Cell matrix associated nucleators in a high supercooling capacity beetle. Cryo-Lett 4:65–71
Block W, Young SR (1979) Measurement of supercooling in small arthropods and water droplets. Cryo-Lett 1:85–91
Chino H (1985) Lipid transport: Biochemistry of hemolymph lipophorin. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry and pharmacology, vol 10, Chap 4. Pergamon Press, Oxford New York Paris, pp 115–135
Danks HV (1978) Modes of seasonal adaptation in insects. I. Winter survival. Can Ent 110:1167–1205
Davis B (1964) Disc electrophoresis. II. Method and application to human serum proteins. Ann NY Acad Sci 121:404–427
Duman JG (1977) The role of macromolecular antifreeze in the darkling beetle,Meracantha contracta. J Comp Physiol 115:279–286
Duman JG, Horwath KL, Tomchaney A, Patterson JL (1982) Antifreeze agents in terrestrial arthropods. Comp Biochem Physiol 73A:545–555
Duman JG, Horwath KL (1983) The role of hemolymph proteins in the cold tolerance of insects. Annu Rev Physiol 45:261–270
Duman JG, Morris JP, Castellino FJ (1984) Purification and composition of an ice nucleating protein from queens of the hornet,Vespula maculata. J Comp Physiol B 154:79–83
Duman JG, Neven LG, Beals JM, Olson KR, Castellino FJ (1985) Freeze-tolerance adaptation, including haemolymph protein and lipoprotein nucleators in the larvae of the craneflyTipula trivittata. J Insect Physiol 31:1–8
Fairbanks G, Stek TL, Wallach DFH (1971) A very intensive staining method useful for minor components and assessment of purity. Biochemistry 10:2606–2617
Gilbert L, Chino H (1974) Transport of lipids in insects. J Lipid Res 15:439–456
Havel RJ, Eder HA, Bragdon JH (1955) The distribution and chemical composition of ultracentrifugally separated lipoproteins in human serum. J Clin Invest 34:1345–1353
MacKenzie AP (1977) Non-equilibrium freezing behavior of aqueous systems. Philos Trans R Soc Lond Ser B 278:167–189
Perkins HF, Aranoff S (1959) A paper chromatographic method for the purification of shikimic acid-u-C14 obtained from culture filtrates of a mutant ofEscherichia coli. Can J Biochem Physiol 37:149–150
Ramsay JA, Brown RH (1955) Simplified apparatus and procedure for freezing point determinations upon small volumes of fluid. J Sci Instrum 32:372–375
Raymond S, Miles JL, Lee JCJ (1966) Lipoprotein patterns on acrylamide gel electrophoresis. Science 151:346–347
Ring RA (1980) Insects and their cells. In: Ashwood-Smith MK, Farrant J (eds) Low Temperature Preservation in Medicine and Biology. University Park Press, Baltimore, Maryland, USA, pp 187–217
Salt RW (1958) Natural occurrence of glycerol in producing abnormally low supercooling and freezing points in an insect,Bracon cephi (Gahan). Nature 181:1281
Salt RW (1966) Factors influencing nucleation in supercooled insects. Can Ent 95:1190–1202
Sömme L (1964) Effects of glycerol on cold hardiness in insects. Can J Zool 44:113–117
Sömme L (1982) Supercooling and winter survival of terristrial arthropods. Comp Biochem Physiol 73A:519–543
Sömme L, Conradi-Larsen EM (1977) Cold hardines of collembolans and oribatid mites from wind swept mountain ridges. Oikos 29:118–126
Thomas KK (1979) Isolation and partial characterization of the hemolymph lipoproteins of the wax moth,Galleria mellonella. Insect Biochem 9:211–219
Zachariassen KE (1982) Nucleating agents in cold hardy insects. Comp Biochem Physiol 73A:557–562
Zachariassen KE (1985) Physiology of cold tolerance in insects. Physiol Rev 65:799–832
Zachariassen KE, Baust JG, Lee RE (1982) A method for quantitative determination of ice nucleating agents in insect hemolymph. Cryobiology 19:180–184
Zachariassen KE, Hammel HT (1976) Nucleating agents in the hemolymph of insects tolerant to freezing. Nature 262:285–287
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Neven, L.G., Duman, J.G., Beals, J.M. et al. Overwintering adaptations of the stag beetle,Ceruchus piceus: removal of ice nucleators in the winter to promote supercooling. J Comp Physiol B 156, 707–716 (1986). https://doi.org/10.1007/BF00692749
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00692749