Summary
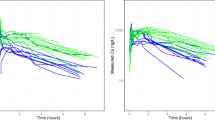
The pharmacokinetics of intravenous ifosfamide were determined in 16 patients with carcinoma of the bronchus. In all 25% (4) of these patients were obese (i.e. >20% over their ideal body weight). The terminal elimination half-life (t1/2 β) was found to be higher in the obese group than in the control group (6.36 h, range 5.77–7.45 h) vs 4.95 h, range 1.82–6.48 h( (P< 0.05). This prolongation of the elimination half-life was due to an increased volume of distribution (Vdβ) in the obese group (42.81 l, range 35.49–51.90 l) vs 33.70 l range (17.76–50.62 l) (P<0.05). There was therefore no significant difference in total plasma clearance between the obese and normal groups. No correlation of ifosfamide plasma half-life was observed with total body weight (TBW) or ideal body weight (IBW). However, a significant positive correlation was observed between the percentage of IBW and plasma half-life. A strong positive correlation was observed between IBW and the plasma clearance of ifosfamide. The Vdβ correlated with both TBW and the percentage of IBW, but not with IBW itself. When Vdβ was normalised for IBW, there was a strong positive correlation with the percentage of IBW, suggesting that ifosfamide distribution into the TBW is higher than that into the IBW.
Similar content being viewed by others
References
Abernethy DR, Greenblatt DJ (1982) Pharmacokinetics of drugs in obesity. Clin Pharmacokinet 7: 108–124
Abernethy DR, Greenblatt DJ (1985) Ibuprofen disposition in obese individuals. Arthritis Rheum 28: 1117–1121
Abernethy DR, Greenblatt DJ, (1986) Drug disposition in obese humans, an update. Clin Pharmacokinet 11: 199–213
Abernethy DR, Greenblatt DJ, Divoll M, Harmatz JS, Shader RI (1981) Alterations in drug distribution and clearance due to obesity. J Pharm Exp Ther 217: 681–685
Abernethy DR, Greenblatt DJ, Divoll M, Shader RI (1983) Enhanced glucuronide conjugation of drugs in obesity; studies of lorazepam, oxazepam and acetaminophen. J Lab Clin Med 101: 873–880
Abernethy DR, Todd EL, Schwartz JB (1985) Caffeine disposition in obesity. Br J Clin Pharmacol 20: 61–66
Bauer LA, Wareing-Tran MS, Drew Edwards WA, Raisys V, Jack R, Dellinger P, Simonowitz D (1985) Cimetidine clearance in the obese. Clin Pharmacol Ther 37: 426–430
Benedek IH, Fiske WD, Griffen WO, Bell RM, Blouin RA, McNamara PJ (1983) Serum alpha-1-acid glycoprotein and the binding of drugs in obesity. Br J Clin Pharmacol 16: 751–754
Brade WP, Herdrich K, Varini M (1985) Ifosfamide — pharmacology, safety and therapeutic potential. Cancer Treat Rev 12: 1–47
Brade W, Seeber S, Herdrich K (1986) Comparative activity of cyclophosphamide and ifosfamide. Cancer Chemother Pharmacol 18 [Suppl 2]: 1–9
Brock N (1983) The oxazaphosphorines. Cancer Treat Rev 10 [Suppl A]: 3–15
Brock N, Hohrst H-J (1963) Über die Aktivierung von Cyclophosphamid in vivo und in vitro. Arzneim Forsch 13: 1021–1031
Bryant BM, Jarman M, Ford HT, Smith IE (1980) Prevention of isophosphamide-induced urothelial toxicity with 2-mercaptoethane sulphonate sodium (mesnum) in patients with advanced carcinoma. Lancet II: 657–659
Connors TA, Cox PJ, Farmer PB, Foster AB, Jarman M (1974) Some studies of the active intermediates formed in the microsomal metabolism of cyclophosphamide and isophosphamide. Biochem Pharmacol 23: 115–129
Costanzi JJ, Gagliano R, Loukas D, Panettiere FJ, Hokanson JA (1978) Ifosfamide in the treatment of recurrent or disseminated lung cancer. A phase II study of two dose schedules. Cancer 41: 1715–1719
Dunkelman SS, Fairhurst B, Plager J (1964) Cortisol metabolism in obesity. J Clin Endocrinol Metab 24: 832–841
Gal P, Jusko WJ, Yurchak AM, Franklin BA (1978) Theophylline disposition in obesity. Clin Pharmacol Ther 23: 438–444
Lind MJ, Margison JM, Cerny T, Thatcher N, Wilkinson PM (1989) Comparative pharmacokinetics and alkylating activity of fractionated oral and intravenous ifosfamide in patients with bronchogenic carcinoma. Cancer Res 49: 753–757
Margison JM, Cerny T, Thatcher N, Wilkinson PM (1986) A simple quantitative HPLC assay for ifosfamide in biological fluids. Biomed Chromatogr 1(3): 101–103
McIntosh JEA, McIntosh RP (1980) Mathematical modeling and computing in endocrinology. Springer, Berlin Heidelberg New York
Milsap RL, Plaisance KI, Jusko WJ (1984) Prednisolone disposition in obese men. Clin Pharmacol Ther 36: 824–831
Powis G (1983) Effects of disease states on pharmacokinetics of anticancer drugs. In: Ames MM, Powis G, Kovach JS (eds) Pharmacokinetics of anticancer agents in humans. Elsevier, Amsterdam, p 63
Powis G, Reece P, Ahmann DL, Ingle JN (1987) Effect of body weight on the pharmacokinetics of cyclophosphamide in breast cancer patients. Cancer Chemother Pharmacol 20: 219–222
Rodvold KA, Rushing DA, Tewksbury DA (1988) Doxorubicin clearance in the obese. J Clin Oncol 6: 1321–1327
Rohrbaugh TM, Danish M, Ragni MC, Yaffe (1982) The effect of obesity on the apparent volume of distribution of theophylline. Pediatr Pharmacol 2: 75–83
Sketris I, Lesar T, Zaske DE, Cipolle RJ (1981) Effect of obesity on gentamicin pharmacokinetics. J Clin Pharmacol 21: 288–293
Silverstone JT, Gordon RP, Stunkard AJ (1969) Social factors in obesity in London. Practitioner 202: 682
Teufel G, Pfleiderer A (1986) Ifosfamid im Vergleich zu Endoxan bei fortgeschrittenen Ovarialkarzinomen. Geburtshilfe Frauenheilkd 36: 274
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lind, M.J., Margison, J.M., Cerny, T. et al. Prolongation of ifosfamide elimination half-life in obese patients due to altered drug distribution. Cancer Chemother. Pharmacol. 25, 139–142 (1989). https://doi.org/10.1007/BF00692355
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00692355