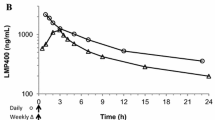
Abstract
Antifolates have been shown to increase the DNA strand breaks produced by the topoisomerase inhibitor etoposide. PT523 is a potent new antifolate that cannot be polyglutamated. Human SCC-25 squamous carcinoma cells were exposed to methotrexate, trimetrexate or PT523 at a concentration of 5 μM for 24 h along with various concentrations of etoposide or novobiocin during the final 2 h. Isobologram analysis of the treatment combinations indicated that exposure of the cells to PT523/etoposide, methotrexate/etoposide, PT523/novobiocin, methotrexate/novobiocin and trimetrexate/novobiocin resulted in greater than additive cytotoxicity. DNA alkaline elution studies with the same drug combinations indicated that there were three- to four-fold increases in the radiation equivalent (rad equivalent) strand breaks in the cellular DNA with etoposide or novobiocin along with the antifolate compared with the topoisomerase II inhibitors alone. Tumor growth delay studies were carried out in the murine SCC VII squamous carcinoma. PT523 (0.5 mg/kg) and methotrexate (2 mg/kg) were administered by 7-day continuous infusion while trimetrexate (3.75 mg/kg) was administered intraperitoneally daily on days 7–9. Etoposide (10 mg/kg) and novobiocin (100 mg/kg) were administered intraperitoneally on alternate days (7, 9, 11). The combinations of PT523 with etoposide or novobiocin were significantly more effective than methotrexate and etoposide or novobiocin, producing tumor growth delays of 8.4 days and 6.9 days, respectively. Overall, the antifolate/topoisomerase II inhibitor treatment combinations produced tumor growth delays that were apparently additive to greater than additive.
Similar content being viewed by others
References
Adachi Y, Luke M, Laemmli UK (1991) Chromosome assembly in vitro: topoisomerase II is required for condensation. Cell 64:137–148.
Barry MA, Behnke CA, Eastman A (1990) Activation of programmed cell death (apoptosis( by cisplatin, other anticancer drugs, toxins and hyperthermia. Biochem Pharmacol 40:2353–2362
Berenbaum MC (1977) Synergy, additivism and antagonism in immunosuppression. A critical review. Clin Exp Immunol 28:1–18
Berrios M, Osheroff N, Fisher PA (1985) In situ localization of DNA topoisomerase II, a major polypeptide component of theDrosophila nuclear matrix fraction. Proc Natl Acad Sci USA 82:4142–4146
Bertino JR, Mini E, Sobrero A, Moroson BA, Love T, Jastreboff M, Carman M, Srimatkandada S, Dube S (1986) Methotrexate resistant cells are targets for selective chemotherapy. Adv Enzyme Regul 24:3–11
Brown JM, Lemmon MJ (1990) Potentiation by the hypoxic cytotoxin SR-4233 of cell killing produced by fractionated irradiation of mouse tumors. Cancer Res 50:7745–7749
Brown JM, Lemmon MJ (1991) Tumor hypoxia can be exploited to preferentially sensitize tumors to fractionated regimen. Int J Radiat Oncol Biol Phys 20:457–461
Catten M, Bresnahan D, Thompson S, Chalkly R (1986) Novobiocin precipitates histones at concentrations normally used to inhibit eukaryotic type II topoisomerase. Nucleic Acids Res 14:3671–3686
Chabner BA (1985) Methoxtrexate. In: Devita VT, Hellman S, Rosenberg SA (eds) Pharmacologic principles of cancer treatment. Lippincott, Philadelphia, pp 229–255
Deen DF, Williams ME (1977) Isobologram analysis of X-ray-BCNU interactions in vitro. Radiat Res 79:483–491
Dewey WC, Stone LE, Miller HH, Giblak RE (1971) Radiosensitization with 5-bromodeoxyuridine of Chinese hamster cells x-irradiated during different phases of the cell cycle. Radiat Res 47:672–688
DiNardo S, Voelkel K, Sternglanz R (1984) DNA toposiomerase II mutant ofSaccharomyces cervisiae: topoisomerase II is required for segregation of daughter molecules at the termination of DNA replication. Proc Natl Acad Sci USA 81:2616–2620
Downes CS, Ord MJ, Mulligan AM, Collins ARS, Johnson RT (1985) Novobiocin inhibition of DNA excision repair may occur through effects on mitochondrial structure and ATP metabolism, not on repair topoisomerases. Carcinogenesis 6:1343–4352
Earnshaw WC, Halligan B, Cooke CA, Heck MMS, Liu LF (1986) Topoisomerase II is a structural component of mitotic chromosome scaffolds. J Cell Biol 100:1706–1715
Eastman A (1993) Apoptosis: a product of programmed and unprogrammed cell death. Toxicol Appl Pharmacol 121:160–164
Eastman A, Barry MA (1992) The origins of DNA breaks: a consequence of DNA damage, DNA repair, or apoptosis? Cancer Invest 10:229–240
Edenberg HJ (1980) Novobiocin inhibition of simian virus 40 DNA replication. Nature 286:529–531
Erba E, Sen S, Lorico A, D'Incalci M (1992) Potentiation of etoposide cytotoxicity against a human ovarian cancer cell line by treatment with non-toxic concentrations of methotrexate or aphidicolin. Eur J Cancer 28:66–71
Fernandes DJ, Danks MK, Beck WT (1990) Decreased nuclear matrix DNA topoisomerase II in human leukemia cells resistant to VM-26 and m-AMSA. Biochemistry 29:4235–4241
Frei E III, Cucchi CA, Rosowsky A, Tantravahi R, Bernal S, Erwin TJ, Ruprecht RM, Haseltine WA (1985) Alkylating agent resistance: in vitro studies with human cell lines. Proc Natl Acad Sci USA 82:2158–2162
Fry DW (1990) Cytotoxic synergism between trimetrexate and etoposide: evidence that trimetrexate potentiates etoposide-induced protein-associated DNA strand breaks in L1210 leukemia cells through alterations in intracellular ATP concentrations. Biochem Pharmacol 40:1981–1988
Gasser SM, Laemmli UK (1986) The organization of chromatin loops: characterization of a scaffold attachment site. EMBO J 5:511–518
Gedick CM, Collins AR (1990) Comparison of effects of fostriecin, novobiocin, and camptothecin, inhibitors of DNA topoisomerases, on DNA replication and repair in human cells. Nucleic Acids Res 18:1007
Gellert M, O'Dea MH, Itoh T, Tomizawa J (1976) Novobiocin growth inhibition, cell killing and DNA lesions in cultured L5178Y cells by the reduction of DNA repair efficiency. Proc Natl Acad Sci USA 73:4474–4478
Golas B, Malec J (1989) Enhancement of methotrexate-induced growth inhibition, cell killing and DNA lesions in cultured L5178Y cells by the reduction of DNA repair efficiency. Biochem Pharmacol 38:1743–1748
Heck MMS, Hittelman WN, Earnshaw WC (1988) Differential expression of topoisomerases I and II during the eukaryotic cell cycle. Proc Natl Acad Sci USA 85:1086–1090
Hickman JA (1992) Apoptosis induced by anticancer drugs. Cancer Metastasis Rev 11:121–139
Jukes TH (1987) Searching for magic bullets: early approaches to chemotherapy, antifolates, metrexate: The Bruce F. Cain Memorial Awards Lecture. Cancer Res 53:3976
Kaufmann SH, Desnoyers S, Ottaviano Y, Davidson NE, Poirier GG (1993) Specific proteolytic cleavage of poly(ADP-ribose) polymerase: an early marker of chemotherapy-induced apoptosis. Cancer Res 53:3976
Lee FYF, Flannery DJ, Siemann DW (1992) Modulation of the cell cycle-dependent cytotoxicity of adriamycin and 4-hydroperoxycyclophosphamide by novobiocin, an inhibitor of mammalian topoisomerase II. Cancer Res 52:3515–3520
Li JC, Kaminskas E (1984) Accumulation of DNA strand breaks and methotrexate cytotoxicity. Proc Natl Acad Sci USA 81:5694–5698
Lin JT, Bertino JR (1987) Trimetrexate: a second genenation folate antagonist in clinical trial. J Clin Oncol 5:2032–2040
Litchfield JT, Wilcoxon FA (1949) A simplified method of evaluating dose-effect experiments. J Pharmacol Exp Ther 96:99
Liu LF (1989) DNA topoisomerase poisons as anti-tumor drugs. Annu Rev Biochem 58:351–375
Lorico A, Toffoli G, Boiocchi M, Erba E, Broggini M, Rappa G, D'Incalci M (1988) Accumulation of DNA strand breaks in cells exposed to methotrexate or N10-propargyl-5,8-dideazafolic acid. Cancer Res 48:2036–2041
Lorico A, Boiocchi M, Rappa G, Sen S, Erba E, D'Incalci M (1990) Increase in topoisomerase-II-mediated DNA breaks and cytotoxicity of VP16 in human U937 lymphoma cells pretreated with low doses of methotrexate. Int J Cancer 45:156–162
Mattern MR, Painter RB (1979) Dependence of mammalian DNA replication on DNA supercoiling. Biochim Biophys Acta 563:306–312
Mattern MR, Paone RF, Day RS III (1982) Eukaryotic DNA repair is blocked at different steps by 8 inhibitors of DNA topoisomerases and of DNA polymerases α and β. Biochim Biophys Acta 697:6–13
Nelson WG, Liu LF, Coffey DS (1986) Newly replicated DNA is associated with DNA topoisomerase II in cultured rat prostatic adenocarcinoma cells. Nature 322:187–189
Paulson JR, Laemmli UK (1977) The structure of histone-depleted metaphase chromosomes. Cell 12:817–828
Rhee MS, Galivan J, Tyobekas EM, Sherman ML, Rosowsky A (1993) Effect of a novel antifolate,Nα-(4-amino-4-deoxypteroyl)-Nδ-hemiphthaloyl-1-ornithine (PT523), on growth of H35 rat hepatoma and HEPG2 human hepatoma cells. In: Ayling JE (ed) Chemistry and biology of pteridines and folates. Plenun Press, New York, pp 461–464
Rhee MS, Galivan J, Wright JE, Rosowsky A (1994) Biochemical studies on PT523, a potent non-polyglutamatable antifolate, in cultured cells. Mol Pharmacol 45:783–791
Rheinwald JG, Beckett MA (1981) Tumorigenic keratinocyte lines requiring anchorage and fibroblast support cultured from human squamous cell carcinomas. Cancer Res 41:1657–1663
Rosowsky A (1989) Chemistry and biological activity of antifolates. Prog Med Chem 26:1–252
Rosowsky A (1992) Development of new antifolate analogs as anticancer agents. Am J Pharm Eduy. 56:453–463
Rosowsky A, Bader H, Cucchi C, Moran RG, Kohler W, Freisham JH (1988) Methotrexate analogues. 33.N δ-Acyl-N δ-(4-amino-4-deoxypteroyl)-l-ornithine derivatives: synthesis and in vitro antitumor activity. J Med Chem 31:1331–1344
Rosowsky A, Bader H, Forsch RA (1989) Synthesis of the folylpolyglutamate synthetase inhibitorNα-pteroyl-l-ornithine and itsN δ-benzoyl andN δ-hemiphthaloyl derivatives, and an improved synthesis ofN δ-(4-amino-4-dexoypteroyl)-N δ-hemiphthaloyl-l-ornithine. Pteridines 1:91–98
Rosowsky A, Bader H, Frei E III (1991) In vitro and in vivo antitumor activity ofNα-(4-amino-4-dexoypteroyl)-N δ-hemiphthaloyl-l-ornithine (PT523), a potent side chain modified aminopterin analog that connot form polyglutamates. Proc Am Assoc Cancer Res 32:325
Rosowsky A, Bader H, Freisham JH (1991) Synthesis and biological activity of methotrexate analogues with two acid groups and a hydrophobic aromatic ring in the side chain. J Med Chem 34:574–579
Ross WE (1985) DNA topoisomerases as targets for cancer therapy. Biochem Pharmacol 34:4191–4195
Sen S, D'Incalci M (1992) Apoptosis: biochemical events and relevance to cancer chemotherapy. FEBS Lett 307:122–127
Sirotnak FM (1987) Determinants of resistance to antifolates: biochemical phenotypes, their frequency of occurrence and circumvention. J Nat Cancer Inst Monogr 5:27–35
Steel GG, Peckman MJ (1979) Exploitable mechanisms in combined radiotherapy-chemotherapy: the concept of additivity. Oncol Biol Phys 15:85–91
Tallarida RJ, Murray RB (1981) Manual of pharmacologic calculations with computer programs. Springer, New York Berlin Heidelberg
Teicher BA, Cucchi CA, Lee JB, Flatow JL, Rosowsky A, Frei E III (1986) Alkylating agents: in vitro studies of cross-resistance patterns. Cancer Res 46:4379–4383
Teicher BA, Holden SA, Kelley MJ, Shea TC, Cucchi CA, Rosowsky A, Henner WD, Frei III E (1987) Characterization of a human squamous carcinoma cell line resistant to cis-diamminedichloroplatinum(II). Cancer Res 47:388–393
Teicher BA, Herman TS, Shulman L, Bubley G, Coleman CN, Frei E III (1991) Combination of etanidazole with cyclophosphamide and platinum complexes. Cancer Chemother Pharmacol 28:153–158
Teicher BA, Holden SA, Herman TS, Alvarez Sotomayor E, Khanekar V, Rosbe KW, Brann TW, Korbut TT, Frei E III (1991) Characteristics of five human tumor cell lines and sublines resistant tocis-diamminedichloroplatinum(II). Int J Cancer 47:252–260
Utsumi H, Shibuya ML, Kosaka T, Buddenbaum WE, Elkind MM (1990) Abrogation by novobiocin of cytotoxicity due to the topoisomerase II inhibitor amsacrine in Chinese hamster cells. Cancer Res 50:2577–2581
Vogelstein B, Pardol DM, Coffey DS (1980) Supercoiled loops and eukaryotic DNA replication. Cell 22:79–85
Wang JC (1985) DNA topoisomerases. Annu Rev Biochem 54:665–697
Wang JC (1987) Recent studies of DNA topoisomerase. Biochim Biophys Acta 909:1–9
Wright JE, Pardo AM, Trites DH, Menon K, Rosowsky A (1993) Pharmacokinetics and antifolate activity ofN δ-(4-amino-4-deoxypteroyl)-N δ-hemiphthaloyl-l-ornithine (PT523) in SCC VII murine squamous cell carcinoma. AACR Proc 34:284
Wu H-Y, Shyy S, Wang JC, Liu LF (1988) Transcription generates positively and negatively supercoiled domains in the template. Cell 53:433–440
Yang L, Wold MS, Li JJ, Kelly TJ, Liu LF (1987) Roles of DNA topoisomerases in simian virus 40 DNA replication. Proc Natl Acad Sci USA 84:950–954
Zeman EM, Hirst VK, Lemmon MJ, Brown JM (1988) Enhancement of radiation-induced tumor cell killing by the hypoxic cell toxin SR 4233. Radiother Oncol 12:209–218
Zwelling LA (1985) DNA topoisomerase II as a target of antineoplastic drug therapy. Cancer Metastasis Rev 4:263–276
Zwelling LA, Kohn KW, Ross WE, Ewig RAG, Anderson T (1978) Kinetics of formation and disappearance of a DNA cross-linking effect in mouse leukemia L1210 cells treated withcis- andtrans-diamminedichloroplatinum(II). Cancer Res 38:1762–1768
Zwelling LA, Anderson T, Kohn KW (1979) DNA-protein and interstrand cross-linking bycis- andtrans-platinum(II) diamminedichloride in L1210 mouse leukemia cells and relation to cytotoxicity. Cancer Res 39:365–369
Zwelling LA, Silverman L, Estey E (1986) Intercalator-induced topoisomerase II-mediated DNA cleavage and its modification by antineoplastic antimetabolites. Int J Radiat Oncol Biol Phys 12:1041–1047
Author information
Authors and Affiliations
Additional information
This work was supported by NIH grants PO1-CA19589 and RO1CA25394
Rights and permissions
About this article
Cite this article
Holden, S.A., Teicher, B.A., Robinson, M.F. et al. Antifolates can potentiate topoisomerase II inhibitors in vitro and in vivo. Cancer Chemother. Pharmacol. 36, 165–171 (1995). https://doi.org/10.1007/BF00689203
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00689203