Abstract
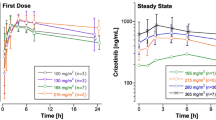
It has often been reported that the oxazaphosphorines ifosfamide and cyclophosphamide induce their own metabolism. This phenomenon was studied in 21 paediatric patients over 35 courses of therapy. All patients received 9 g m−2 of ifosfamide as a continuous infusion over 72 h. Plasma concentrations of parent drug and of the major metabolite in plasma, 3-dechloroethylifosfamide (3DC) were determined using a quantitative thin-layer chromatography (TLC) technique: A one-compartment model was fitted simultaneously to both ifosfamide and 3DC data. The model included a time-dependent clearance term, increasing asymptotically from an initial value to a final induced clearance and characterised by a first-order rate constant. A time lag, before induction of clearance began, was determined empirically. Metabolite kinetics were characterised by an elimination rate constant for the metabolite and a composite parameter comprising a formation clearance, proportional to the time-dependent clearance of parent drug, divided by the volume of distribution of the metabolite. Thus, the parameters to estimate were the volume of distribution of parent drug (V), initial clearance (Cli), final clearance (Cls), the rate constant for changing clearance (Kc), the elimination rate constant for the metabolite (Km) and Vm/fm, the metabolite volume of distribution divided by the fractional clearance to 3DC. The model of drug and metabolite kinetics produced a good fit to the data in 22 of 31 courses. In a further 4 courses an auto-inductive model for parent drug alone could be used. In the remaining courses, auto-induction could be demonstrated, but there were insufficient data to fit the model. For some patients this was due to a long time lag (up to 54 h) relative to the infusion time. The time lag varied from 6 to 54 (median, 12) h and values for the other parameters were Cli, 3.27±2.52 l h−1m−2, Cls, 7.50±3.03 l h−1m−2, V, 22.0±11.0 l m−2,Kc, 0.086±0.074 h−1;Km, 0.159±0.077 h−1 and Vm/fm, 104±82 l m−2. The values ofKc correspond to a halflife of change in clearance ranging from 2 to 157 h, although for the majority of the patients the half-life was less than 7 h and a new steady-state level was achieved during the 72 h infusion period. This model provides insight into the time course of enzyme induction during ifosfamide administration, which may continue for up to 10 days in some protocols. Since other drugs, including common anti-neoplastic agents, are metabolised by the same P450 enzyme as is ifosfamide, auto-induction may have implications for the scheduling of combined chemotherapy.
Similar content being viewed by others
References
Abramson FP (1986) Kinetic models of induction. I. Persistence of the inducing substance. J Pharm Sci. 75:223–228
Abramson FP (1986) Kinetic models of induction. II. Decreased turnover of a product or its prescursor. J Pharm Sci 75:229–232
Adamson PC, Boylan JF, Balis FM, Murphy RF, Godwin KA, Gudas LJ, Poplack DG (1993) Time course of induction of metabolism ofall-trans-retinoic acid and the up-regulation of cellular retinoic acid-binding protein. Cancer Res 53:472–476
Boddy AV, Idle JR (1992) Combined thin-layer chromatography-photography-densitometry for the quantification of ifosfamide and its principal metabolites in urine, cerebrospinal fluid and plasma. J Chromatogr Biomed Appl 575:137–142
Boddy AV, Wyllie R, Price L, Pearson ADJ, Idle JR (1992) Increased clearance of ifosfamide in children during continuous infusion or repeated bolus administration. Pharm Res 9:S314
Boddy AV, Yule SM, Wyllie R, Price L, Pearson ADJ, Idle JR (1993) Pharmacokinetics and metabolism in children of ifosfamide administered as a continuous infusion. Cancer Res 53: 3758–3764
Boddy AV, Yule SM, Wyllie R, Price L, Pearson ADJ, Idle JR (1994) Variation of ifosfamide pharmacokinetics and metabolism following repeated administration. Proc Amer Assoc Cancer Res 35:246
Chang S-L, Emmick K, Wedlund PJ (1985) Characterization of antipyrine autoinduction in the rat utilizing a new microsampling technique for serial blood sample collections. J Pharm Sci 75:456–458
Chang TKH, Waxman DJ (1993) Cyclophosphamide modulates rat hepatic cytochrome P450 2C11 and steroid 5α-reductase activity and meassenger RNA levels through the combined action of acrolein and phosphoramide mustard. Cancer Res 53: 2490–2497
Chang TKH, Weber GF, Crespi CL, Waxman DJ (1993) Differential activation of cyclophosphamide and ifosphamide by cytochromes P-450 2B and 3A in human liver microsomes. Cancer Res 53:5629–5637
Denlinger CL, Stryker KK, Slusher LB, Vesell ES (1987) Studies on theophylline metabolism: autoinduction and inhibition by antipyrine. Clin. Pharmacol Ther 41:522–530
D'Incalci M, Bolis G, Facchinetti T, Mangioni C, Morasca L, Morazzoni P, Salmona M (1979) Decreased half life of cyclophosphamide in patients under continual treatment. Eur J Cancer 13:7–10
Donelli MG, Garattini S (1971) Drug metabolism after repeated treatments with cytotoxic agents. Eur J Cancer 7:361–364
Faber OK, Mouridsen HT, Skovsted L (1974) The biotransformation of cyclophosphamide in man: influence of prednisone. Acta Pharmacol Toxicol (copenh) 35:195–200
Fasola G, Lo Greco P, Calori E, Zilli M, Verlicchi F, Rosa Motta M, Ricci P, Baccarani M, Tura S (1991) Pharmacokinetics of high-dose cyclophosphamide for bone marrow transplantation. Haematologica (Pavia) 76:120–125
Gibaldi M, Perrier D (1982) Pharmacokinetics. in: Swarbrick J (ed) Pharmacokinetics, 2nd edn, vol. 15. Marcel Dekker, New York, 303–307
Goren MP, Wright RK, Pratt CB, Pell FE (1986) Dechloroethylation of ifosfamide and neurotoxicity. Lancet II: 1219–1220
Graham MI, Shaw IC, Souhami RL, Sidau B, Harper PG, McLean AEM (1983) Decreased plasma half-life of cyclophosphamide during repeated high-dose administration. Cancer Chemother Pharmacol 15:192–193
Houston JB (1981) Drug metabolite kinetics. Pharmacol Ther 15:521–552
Kudriakova TB, Sirota LA, Rozova GI, Gorkov VA (1992) Autoinduction and steady-state pharmacokinetics of carbamazepine and its major metabolites. Br J Clin Pharmacol 33: 611–615
Kurowski V, Wagner T (1993) Comparative pharmacokinetics of ifosfamide, 4-hydroxyifosfamide, chloroacetaldehyde and 2-and 3-dechloroethylifosfamide in patients on fractionated intravenous ifosfamide therapy. Cancer Chemother Pharmacol 33:36–42
Leblane BA, Waxman DJ (1990) Mechanisms of cyclophosphamide action on hepatic p-450 expression. Cancer Res 50: 5720–5726
Lind MJ, Roberts HL, Thatcher N, Idle JR (1990) The effect of route of administration and fractionation of dose on the metabolism of ifosfamide. Cancer Chemother Pharmacol 26:105–111
Ludden T (1991) Nolinear pharmacokinetics. Clinical implications. Clin Pharmacokinet 20:429–446
MacPhee GJA, Thompson GG, Scobie G, Agnew E, Park BK, Murray T, McColl KEL, Brodie MJ (1984) Effects of cimetidine on carbamazepine auto-and hetero-induction in man. Br J Clin Pharmacol 18:411–419
McClure MT, Stupans I (1992) Investigation of the mechanism by which cyclophosphamide alters cytochrome P450 in male rats. Biochem Pharmacol 43:2655–2658
Miller AA, Miller BE, Hoffken K, Schmidt CG (1987) Clinical pharmacology of aminoglutethimide in patients with metastatic breast cancer. Cancer Chemother Pharmacol 20:337–341
Philip PA, Harper PG, Rogers HJ (1989) Effect of cancer chemotherapy on dapsone N-acetylation in man. Cancer Chemother Pharmacol 23:395–396
Pitlick WH, Levy RH, Troupin AS, Green JR (1976) Pharmacokinetic model to describe self-induced decreases in steadystate concentration of carbamazepine. J Pharm Sci 65: 462–463
Relling MV, Evans R, Dass C, Desiderio DM, Nemec J (1992) Human cytochrome P450 metabolism of teniposide and etoposide. J Pharmacol Exp Ther 261:491–496
Skinner R, Sharkey IM, Pearson ADJ, Craft AW (1993) Ifosfamide, mesna and nephrotoxicity in children. J Clin Oncol 11: 173–190
Sladek N (1988) Metabolism of oxazaphosphorines. Pharmacol Ther 37:301–355
Tasso MJ, Boddy AV, Price L, Wyllie RA, Pearson ADJ, Idle JR (1992) Pharmacokinetics and metabolism of cyclophosphamide in paediatric patients. Cancer Chemother Pharmacol 30: 207–211
Walker D, Flinois J-P, Monkman SC, Beloc C, Boddy AV, Cholerton S, Daly AK, Lind MJ, Pearson ADJ, Beaune PH, Idle JR (1994) Identification of the major human hepatic cytochrome P450 involved in activation andN-dechloroethylation of ifosfamide. Biochem. Pharmacol. 47:1157–1163
Zhou X-J, Zhou-Pan X-R, Gauthier T, Placici M, Maurel P, Rahmani R (1993) Human liver microsomal cytochrome P450 3A isozymes mediated vindesine biotransformation. Biochem Pharmacol 45:853–861
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boddy, A.V., Cole, M., Pearson, A.D.J. et al. The kinetics of the auto-induction of ifosfamide metabolism during continuous infusion. Cancer Chemother. Pharmacol. 36, 53–60 (1995). https://doi.org/10.1007/BF00685732
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00685732