Abstract
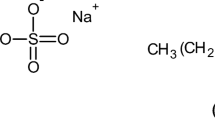
The surface tensions and the phase equilibria of dilute aqueous cationic starch (CS)/surfactant systems were investigated. The degree of substitution of the CS varied from 0.014 to 0.772. The surfactants investigated were sodium dodecyl sulphate (SDS), potassium octanoate (KOct), potassium dodecanoate (KDod) and sodium oleate (NaOl). The concentrations of CS were 0.001, 0.01 and 0.1 w%.
Critical association concentrations (cac) occur at surfactant concentrations well below the critical micelle concentrations of the surfactants, except for KOct, KDod and NaOl at the lowest CS concentrations investigated (0.001 w%). The surface tensions of CS/surfactant solutions decrease strongly already below the cac. This is attributed to the formation of surface active associates by ion condensation. Associative phase separation of gels formed by CS and surfactant takes place at extremely low concentrations when the surfactant/polymer charge ratio is somewhat larger than 1. The gel is higly viscous and contains 40–60% water, depending on the concentration of electrolyte, the surfactant hydrocarbon chain length and the nature of the polar head of the surfactant.
The concentration at which the phase separation occurs decreases with increasing surfactant chain length and the concentration of simple electrolyte, factors that promote micelle formation. This indicates that the gels are formed by association of CS to surfactant micelles. When surfactant well in excess of charge equivalence is added, the gels dissolve because the CS/surfactant complexes acquire a high charge.
Similar content being viewed by others
References
Goddard ED, Ananthapadmanadhan KP (1993) (eds) Interactions of Surfactants with Polymers and Proteins, CRC Press
Hayakawa K, Kwak JCT (1991) In: Rubingh DN, Holland PM (eds) Cationic Surfactants. Marcel Dekker, New York, 2nd ed
Saito S (1991) In: Schick MJ (ed) Nonionic Surfactants. Marcel Dekker, New York, p 189
Robb ID (1981) In: Lucassen-Reynders E (ed) Anionic Surfactants — Physical Chemistry of Surfactant Action, Surfactant Sci Ser, Vol 11, Marcel Dekker, New York, Chap 3
Lindman B, Piculell L (1992) Adv Coll Interface Sci 41:149
Goddard ED, Hannan RB (1976) J Colloid Interface Sci 55:73
Goddard ED, Hannan RB (1977) J Am Oil Chem Soc 54:561
Mukerjee P, Mysels K (1971) Critical Micelle Concentrations of Aqueous Surfactant Systems, NSRDS-NBS 36. National Bureau of Standards, Washington
Huh C, Mason SG (1975) Colloid Polymer Sci 253:566
Simister EA, Thomas RK, Penfold J, Aveyard R, Blinks BP, Cooper P, Fletcher PDI, Lu JR, Sokolowski A (1992) J Phys Chem 96:1383
Bradstreet RB (1965) The Kjeldahl Method for Organic Nitrogen, Academic Press, New York
Manning GS (1978) Q Rev Biophys 11:179
Hayakawa K, Kwak JCT (1983) Macromolecules 16:1642
Thalberg K, Lindman B, Karström G (1991) J Phys Chem 95:6004
Thalberg K, Lindman B (1991) Langmuir 7:277
Flory PJ (1953) Principles of polymer chemistry, Cornell University Press; Ithaca, New York
Thalberg K, Lindman B, Karlström G (1990) J Phys Chem 94:4289
Goddard ED (1986) Colloids Surf 19:301
Thalberg K, Lindman B, Bergfeldt K (1991) Langmuir 7:2893
Shirahama K, Himuro A, Takisawa N (1987) Colloid Polymer Sci 265:96
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Merta, J., Stenius, P. Interactions between cationic starch and anionic surfactants. Colloid Polym Sci 273, 974–983 (1995). https://doi.org/10.1007/BF00660376
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00660376