Abstract
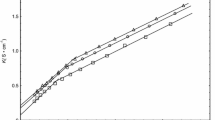
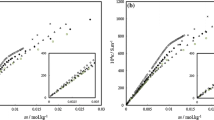
Densities, heat capacities and enthalpies of dilution at 25°C and osmotic coefficients at 37°C were measured for N-octyl- and N-dodecylnicotinamide chlorides in water over an extended concentration region. Partial molar volumes, heat capacities, relative enthalpies and nonideal free energies and entropies at 25°C were derived as a function of the surfactant concentration. For both surfactants, plots of volumes, enthalpies and free energies vs. concentration are regular whereas those of heat capacities and entropies present anomalies at about 0.8 and 0.1m for the octyl and dodecyl compounds, respectively. Changes in the slope of a plot of osmotic coefficients times molality vs. molality were also observed at these same concentrations. These peculiarities are ascribed to micelle structural transitions. The nonideal free energies do not seem to depend on the alkyl chain length when they are plotted vs. m/C cmc . Also, a plot of the nonideal free energy vs. logm/C cmc is roughly independent of the nature of the surfactant because of the constant activity of surfactants in micellar solutions. Nonideal free energies, enthalpies and entropies have been calculated at 15 and 35°C. At each concentration the nonideal free energy is temperature independent as a result of a compensatory effect between enthalpy and entropy. The thermodynamic functions of micellization were graphically evaluated on the basis of the pseudo-phase transition model. These data suggest that the nicotinamide group possesses less hydrophilic character than the ammonium group.
Similar content being viewed by others
References
“Colloids, and Surfactants: Fundamentals, and Applications,” E. Barni and E. Pelizzetti, eds.,Soc. Chim. It., Ann. Chim. (Rome),77, 209 (1987).
The Pyridine Nucleotide Coenzymes, J. Everse, B. Anderson, and K. S. You, eds., (Academic Press, New York, 1982).
K. Kalyanasundaram, T. Colassis, R. Humphry-Baker, P. Savarino, E. Barni, E. Pelizzetti, and M. Gratzel,J. Am. Chem. Soc. in press.
K. Kalyanasundaram, T. Colassis, R. Humphry-Baker, P. Savarino, E. Barni, E. Pelizzetti, and M. Gratzel,J. Coll. Interface Sci. in press.
M. F. Stimson,Am. J. Phys. 23, 614 (1955).
G. S. Kell,J. Chem. Eng. Data 12, 66 (1967).
C. Jolicoeur, and G. Lacroix,Can. J. Chem. 54, 624 (1976).
J. E. Desnoyers, R. De Lisi, C. Ostiguy, and G. Perron, inSolution Chemistry of Surfactants, Vol. 1, K. L. Mittal ed., (Plenum, New York, 1979).
R. De Lisi, S. Milioto, and R. Triolo,J. Solution Chem. 17, 673 (1988).
G. Roux-Desgranges, A. H. Roux, and A. Viallard,J. Chim. Phys. 82, 441 (1985).
F. Quirion and J. E. Desnoyers,J. Colloid Interface Sci. 112, 565 (1986).
R. De Lisi, E. Fisicaro, and S. Milioto,J. Solution Chem. 17, 1015 (1988).
S. Backlund, H. Hoiland, O. J. Kvammen, and E. Ljosland,Acta Chem. Scand. A 36, 698 (1982).
R. De Lisi, C. Ostiguy, G. Perron, and J. E. Desnoyers,J. Colloid Interface Sci. 71, 147 (1979).
L. V. Dearden and E. M. Woolley,J. Phys. Chem. 91, 2404 (1987).
R. De Lisi, G. Perron, and J. E. Desnoyers,Can. J. Chem. 59, 1865 (1981)
J. E. Desnoyers, G. Caron, R. De Lisi, D. Roberts, A. H. Roux, and G. Perron,J. Phys. Chem. 87, 1397 (1983).
R. De Lisi, G. Perron, and J. E. Desnoyers,Can. J. Chem. 58, 959 (1980).
J. E. Desnoyers, R. De Lisi, and G. Perron,Pure and Appl. Chem. 52, 433 (1980).
M. T. Bashford and E. M. Woolley,J. Phys. Chem. 89, 3173 (1985).
E. M. Woolley, private communication.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Lisi, R., Fisicaro, E., Milioto, S. et al. Thermodynamic properties of N-octyl- and N-dodecylnicotinamide chlorides in water. J Solution Chem 19, 247–270 (1990). https://doi.org/10.1007/BF00650457
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00650457