Abstract
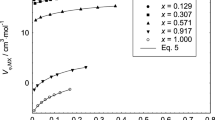
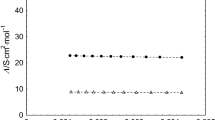
Previously developed additivity schemes for nonelectrolytes have been used to estimate\(\bar V_2^{\text{o}} \) and\(\bar C_{{\text{p}},2}^{\text{o}} \) for tetraalkyl and tetraphenyl methanes in methanol and water. Corrections have been applied to the thermodynamic values of these model compounds to account for a variation in size of the central atom, and these were used to ascertain the effect of charge on\(\bar V_2^{\text{o}} \) and\(\bar C_{{\text{p}},2}^{\text{o}} \) of alkyl and phenyl quaternary ions having N, P and B as central atoms. Investigations of R4NBr, (R=methyl to heptyl) salts show that the charge effect on\(\bar V_2^{\text{o}} \) and\(\bar C_{{\text{p}},2}^{\text{o}} \) of R4N+ ions is large and relatively independent of ion size suggesting that the solvent molecules penetrate the ions. The ability to estimate\(\bar V_2^{\text{o}} \) and\(\bar C_{{\text{p}},2}^{\text{o}} \) of the quaternary ions in the bromide salt solutions has made it possible to make ionic assignments with some confidence;\(\bar V_2^{\text{o}} \) (Br−) has been evaluated as 19.7±2 and 30.2±7 cm3-mol−1 and\(\bar C_{{\text{p}},2}^{\text{o}} \) (Br−) as −83±7 and −68±30 J-K−1-mol−1 in methanol and water, respectively. The use of organic ions for making ionic assignments of\(\bar V_2^{\text{o}} \) and\(\bar C_{{\text{p}},2}^{\text{o}} \) is critically examined and comparisons with other assignments are made. The scaled particle theory is employed to divide the heat capacities of electrolytes into cavity and interaction contributions.
Similar content being viewed by others
References
B. E. Conway,J. Solution Chem. 7, 721 (1978).
R. Zana, G. Perron, and J. E. Desnoyers,J. Solution Chem. 8, 729 (1979).
F. J. Millero,J. Phys. Chem. 75, 280 (1971).
J. I. Kim,J. Phys. Chem. 82, 191 (1978).
M. R. J. Dack, K. J. Bird, and A. J. Parker,Aust. J. Chem. 28, 955 (1975).
R. Zana, G. A. Lage, and C. M. Criss,J. Solution Chem. 9, 667 (1980).
C. Jolicoeur and J. C. Mercier,J. Phys Chem. 81, 1119 (1977).
B. E. Conway, R. Verrall, and J. E. Desnoyers,Trans. Faraday Soc. 62, 2738 (1966).
C. Shin, I. Worsley, and C. M. Criss,J. Solution Chem. 5, 867 (1976).
C. Shin and C. M. Criss,J. Solution Chem. 7, 205 (1978).
Y. S. Choi and C. M. Criss,Faraday Disc. Chem. Soc. 64, 204 (1978).
C. Jolicoeur, P. R. Philip, G. Perron, P. A. Leduc, and J. E. Desnoyers,Can. J. Chem. 50, 3167 (1972).
B. Conway, R. Verrall, and J. E. Desnoyers,Z. Phys. Chem. (Leipzig). 230, 157 (1965).
B. Conway, J. E. Desnoyers, and R. VerrallJ. Phys. Chem. 75, 3031 (1971).
B. S. Krumgalz,J.C.S. Faraday Trans. I 76, 1887 (1980).
R. N. French and C. M. Criss,J. Solution Chem. 10, 231 (1981).
R. N. French and C. M. Criss,J. Solution Chem. 10, 699 (1981).
R. N. French and C. M. Criss,J. Solution Chem. 10, 713 (1981).
O. Redlich and D. M. Meyer,Chem. Rev. 64, 221 (1964).
A. J. Pasztor and C. M. Criss,J. Solution Che. 7, 27 (1978).
S. W. Benson, F. R. Cruickshank, D. M. Golden, G. R. Haugen, H. E. O'Neal, A. S. Rodgers, R. Shaw, and R. Walsh,Chem. Rev. 69, 279 (1969).
J. T. Edward,J. Chem. Educ. 47, 261 (1970).
A. Bordi,J. Phys. Chem. 68, 441 (1964).
B. E. Conway, D. M. Novak, and L. LaliberteJ. Solution Chem. 3, 683 (1974).
C. Shin and C. M. Criss, unpublished results.
F. Kawaizumi and R. Zana,J. Phys. Chem. 78, 1099 (1974).
K. M. Kale and R. Zana,J. Solution Chem. 6, 733 (1977).
F. Kawaizumi and R. Zana,J. Phys. Chem. 78, 627 (1974).
C. Jolicoeur and P. Philip,J. Solution Chem. 4, 3 (1975).
K. Takaizumi and T. Wakabayashi,J. Solution Chem. 9, 809 (1980).
F. J. Millero,J. Phys. Chem. 73, 2417 (1969).
J. Padova and I. Abrahhamer,J. Phys. Chem. 71, 2112 (1967).
F. J. Millero,Chem. Rev. 71, 147 (1971).
G. Perron, N. Desrosiers, and J. E. Desnoyers,Can. J. Chem. 54, 2163 (1976).
J. E. Desnoyers, C. DeVisser, G. Perron, and P. Picker,J. Solution Chem. 5, 605 (1976).
R. Zana and E. Yeager,J. Phys. Chem. 71, 521 (1967).
C. M. Criss and J. W. Cobble,J. Am. Chem. Soc. 86, 5390 (1964).
R. M. Noyers,J. Am. Chem. Soc. 86, 971 (1974).
P. R. Tremaine, N. H. Sagert, and G. J. Wallace,J. Phys. Chem. 85, 1977 (1981).
J. W. Akitt,J.C.S. Faraday Trans. I. 76, 225 (1980).
A. M. Couture and K. J. Laidler,Can. J. Chem. 34, 1210 (1956).
L. G. Hepler,J. Am. Chem. Soc. 61, 1426 (1957).
S. Cabani, G. Conti, and E. Matteoli,Z. Phys. Chem. N. F. 115, 121 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
French, R.N., Criss, C.M. Effect of charge on the standard partial molar volumes and heat capacities of organic electrolytes in methanol and water. J Solution Chem 11, 625–648 (1982). https://doi.org/10.1007/BF00650396
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00650396