Summary
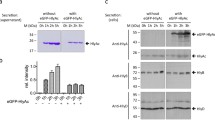
Alkaline phosphatase (AP) is secreted into the medium when the carboxy-terminal 25 amino acids are replaced by the 60 amino acid carboxy-terminal signal peptide (HlyAs) ofEscherichia coli haemolysin (HlyA). Secretion of the AP-HlyAs fusion protein is dependent on HlyB and HlyD but independent of SecA and SecY. The efficiency of secretion by HlyB/HlyD is decreased when AP carries its own N-terminal signal peptide. Translocation of this fusion protein into the periplasm is not observed even in the absence of HlyB/HlyD. The failure of the Sec export machinery to transport the latter protein into the periplasm seems to be due in part to the loss of the carboxy-terminal sequence of AP since even AP derivatives which do not carry the HlyA signal peptide but lack the 25 C-terminal amino acids of AP are localized in the membrane but not translocated into the periplasm.
Similar content being viewed by others
References
Boidol W, Simonis M, Töpert M, Siewert G (1982) Recombinant plasmids with genes for the biosynthesis of alkaline phosphatase ofEscherichia coli. Mol Gen Genet 185:510–512
Collier DN, Bankaitis VA, Weiss JA, Bassford PJ (1988) The antifolding activity of SecB promotes the export of theE. coli maltose-binding protein. Cell 53:273–283
Felmlee T, Pellett S, Welch RA (1985) Nucleotide sequence of anEscherichia coli chromosomal hemolysin. J Bacteriol 163:94–105
Fitts R, Reuveny Z, Amsterdam J, Mulholland J, Botstein D (1987) Substitution of tyrosine for either cysteine in β-lactamase prevents release from the membrane during secretion. Proc Natl Acad Sci USA 84:8540–8543
Garwin JL, Beckwith J (1982) Secretion and processing of ribose-binding protein inEscherichia coli K-12. J Bacteriol 149:789–792
Härtlein M, Schießl S, Wagner W, Rdest U, Kreft J, Goebel W (1983) Transport of hemolysin byEscherichia coli. J Cell Biochem 22:87–97
Hess J (1990) PhD Thesis, University of Würzburg
Hess J, Wels W, Vogel M, Goebel W (1986) Nucleotide sequence of a plasmid-encoded hemolysin determinant and its comparison with a corresponding chromosomal hemolysin sequence. FEMS Microbiol Lett 34:1–11
Holland IB, Wang R, Seror SJ, Blight M (1989) Haemolysin secretion and other protein translocation mechanisms in gram-negative bacteria, In: Baumberg S, Huster I, Rhodes M (eds) In: Microbial products: New approaches. Cambridge University Press, pp 219–254
Inouye H, Michaelis S, Wright A, Beckwith J (1981) Cloning and restriction mapping of the alkaline phosphatase structural gene (phoA) ofEscherichia coli and generation of deletion mutants in vitro. J Bacteriol 146:668–675
Inouye H, Barnes W, Beckwith J (1982) Signal sequence of alkaline phosphatase ofEscherichia coli. J Bacteriol 149:434–439
Ito K, Beckwith J (1981) Role of the mature protein sequence of maltose-binding protein in its secretion across theE. coli cytoplasmic membrane. Cell 25:143–150
Ito K, Wittekind M, Nomura M, Shiba K, Yura T, Muira A, Nashimoto H (1983) A temperature-sensitive mutant ofE. coli exhibiting slow processing of exported proteins. Cell 32:782–797
Josefsson LG, Randall LL (1981) Different exported proteins inE. coli show differences in the temporal mode of processingin vivo. Cell 25:151–157
Kasukawa N, Yura T, Ueguchi C, Akiyama Y, Ito K (1989) Effects of mutations in heat-shock genesgroES andgroEL on protein export inEscherichia coli. EMBO J 8:3517–3521
Koronakis V, Koronakis E, Hughes C (1989) Isolation and analysis of the C-terminal signal directing export ofEscherichia coli hemolysin protein across both bacterial membranes. EMBO J 8:595–605
Koshland D, Botstein D (1980) Secretion of betalactamase requires the carboxy end of the protein. Cell 20:749–760
Koshland D, Botstein D (1982) Evidence for posttranslational translocation of β-lactamase across the bacterial inner membrane. Cell 30:893–902
Kumamoto CA, Beckwith J (1985) Evidence for specificity at a early step in protein export inEscherichia coli. J Bacteriol 163:267–274
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the heads of bacteriophage T4. Nature 227:680–685
Lee C, Beckwith J (1986) Cotranslational and posttranslational protein translocation in prokaryotic systems. Annu Rev Cell Biol 2:315–336
Ludwig A, Vogel M, Goebel W (1987) Mutations affecting activity and transport of haemolysin inEscherichia coli. Mol Gen Genet 206:238–245
Mackman N, Baker K, Gray L, Haigh R, Nicaud J-M, Holland T (1987) Release of a chimeric protein into the medium from Escherichia coli using the C-terminal secretion signal of haemolysin. EMBO J 6:2835–2841
Manoil C, Beckwith J (1985) TnphoA: A transposon probe for protein export signals. Proc Natl Acad Sci USA 82:8129–8133
Minsky A, Summers RG, Knowles JR (1986) Secretion of β-lactamase into the periplasm ofEscherichia coli: evidence for a distinct release step associated with a conformational change. Proc Natl Acad Sci USA 83:4180–4184
Neu HC, Heppel LA (1965) The release of enzymes fromEscherichia coli by osmotic shock and during formation of spheroplasts. J Biol Chem 240:3685–3692
Nicaud JM, Mackman N, Gray L, Holland B (1986) The C-terminal 23 KD peptide ofEscherichia coli haemolysin 2001 contains all the information necessary for its secretion by the haemolysin (Hly) machinery. FEBS Lett 204:331–335
Oliver D, Beckwith J (1981)E. coli mutant pleiotropically defective in the export of secreted proteins. Cell 25:765–772
Pugsley AP, Schwartz M (1985) Export and secretion of proteins by bacteria. FEMS Microbiol Rev 32:3–38
Randall LL, Hardy SJS, Thom JR (1987) Export of proteins: a biochemical view. Annu Rev Microbiol 41:507–541
Robinson A, Austen B (1987) The role of topogenic sequences in the movement of proteins through membranes. Biochem J 246:249–261
Towbin H, Staehlin H, Godron J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Wagner W, Vogel M, Goebel W (1983) Transport of hemolysin across the outer membrane ofE. coli requires two functions. J Bacteriol 154:200–210
Wolfe BP, Rice M, Wickner W (1985) Effects of twosec genes on protein assembly into the plasma membrane ofEscherichia coli. J Biol Chem 260:1836–1841
Author information
Authors and Affiliations
Additional information
Communicated by E.K.F. Bautz
Rights and permissions
About this article
Cite this article
Gentschev, I., Hess, J. & Goebel, W. Change in the cellular localization of alkaline phosphatase by alteration of its carboxy-terminal sequence. Mol Gen Genet 222, 211–216 (1990). https://doi.org/10.1007/BF00633820
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00633820