Abstract
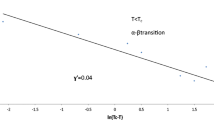
Measurements have been made in the temperature range from 1.3° K to about 71°K with particular attention to the behavior at very low temperatures, and in the neighborhood of the α-β transition at about 23.89° K. Assuming that near 0° K each molecule moves as a single mass point with the passage of lattice waves, the effective Debye temperature at 0° K is extrapolated to be 104±2° K. As the temperature rises above 10° K, the specific heat rises more rapidly than a reasonable Debye model would predict, suggesting the appearance of additional degrees of freedom, which are thought to be the superposition of a librational motion of the molecules superposed on the longitudinal and transverse lattice waves controlling the motion of a molecule's center of mass. The specific heat shows a very sharp high “spike” at the α-β transition with an entropy change of aboutR ln 1.65; there is no evidence for a latent heat associated with this transition.
Similar content being viewed by others
References
C. S. Barrett, L. Meyer, and J. Wasserman,J. Chem. Phys. 47, 592 (1967).
C. S. Barrett and L. Meyer,Phys. Rev. 160, 694 (1967).
A. Eucken,Verhandl. Deut. Physik. Ges. 18, 4 (1916).
K. Clusius,Z. Physik. Chem. B3, 41 (1929).
W. F. Giauque and H. L. Johnston,J. Am. Chem. Soc. 51, 2300 (1929).
H. J. Hoge,J. Res. Natl. Bur. Std. 44, 321 (1950).
C.-H. Fagerstroem and A. C. Hollis Hallett,Ann. Acad. Sci. Fennicae AV1, 210 (1966).
C.-H. Fagerstroem and A. C. Hollis Hallett, to be published.
M. O. Kostriukova and P. G. Strelkov,Dokl. Akad. Nauk SSSR 90, 525 (1953).
M. O. Kostriukova,Soviet Phys.—JETP 3, 771 (1956).
R. H. Beaumont, H. Chihara, and J. A. Morrison,Proc. Phys. Soc. (London)78, 1462 (1961).
B. R. Cairns and G. C. Pimental,J. Chem. Phys. 43, 3432 (1965).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fagerstroem, C.H., Hollis Hallett, A.C. The specific heat of solid oxygen. J Low Temp Phys 1, 3–12 (1969). https://doi.org/10.1007/BF00628329
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00628329