Summary
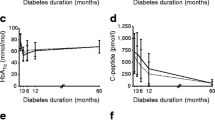
An increased proinsulin to C-peptide molar ratio at the onset of Type 1 (insulin-dependent) diabetes mellitus has been suggested. We studied fasting proinsulin levels and proinsulin/C-peptide ratios in the newly diagnosed diabetic subjects participating in the Canadian/European placebo controlled cyclosporin study at entry, during the one year treatment period and six months of follow-up. Available entry data from 176 out of the 188 allocated patients were compared to 60 age and weight matched control subjects. Fasting proinsulin was significantly elevated in male patients compared to male control subjects (p<0.01), whereas the levels only tended to be elevated in female patients. The proinsulin/C-peptide ratio was three to fourfold elevated in the diabetic groups of both sexes, (p<0.001). Further, proinsulin and C-peptide were studied in 83 cyclosporin and 86 placebo-treated subjects during the trial and follow-up. An additional increase of proinsulin/C-peptide ratio was observed during the first three months of placebo treatment. It remained constantly high for nine months and then declined to entry level. This pattern was not seen in the cyclosporintreated group, where the ratio was unchanged during the 12 months trial and follow-up. The effect of cyclosporin on the induction of non-insulin requiring remission was unrelated to fasting and glucagon stimulated C-peptide levels at entry, whereas 64% of the cyclosporin-treated against 28% of the placebo-treated subjects (p<0.01) went into remission if the proinsulin/C-peptide ratio at entry was above 0.024. If the ratio was below 0.024 at entry, 42% and 33% went into non-insulin requiring remission, respectively (NS). We conclude that fasting proinsulin to C-peptide molar ratio is elevated at the onset of Type 1 diabetes mellitus. A further plateaushape elevation lasting nine months was seen during the remission period. Cyclosporin seems to inhibit or delay this development. The proinsulin/C-peptide ratio at diagnosis may show to be of value in the prediction of remission during cyclosporin treatment.
Article PDF
Similar content being viewed by others
References
Faber OK, Binder C (1977) B-cell function and blood glucose control in insulin-dependent diabetics within the first month of insulin treatment. Diabetologia 13: 263–268
Ludvigsson J, Heding L (1978) Beta-cell function in children with diabetes. Diabetes 27 [Suppl. 1]: 230–234
Agner T, Damm P, Binder C (1987) Remission in IDDM: prospective study of basal C-peptide and insulin dose in 268 consecutive patients. Diabetes Care 10: 164–169
Schiffrin A, Suissa S, Poussier P, Guttmann R, Weitzner G (1988) Prospective study of predictors of β-cell survival in type 1 diabetes. Diabetes 37: 920–925
Wallensteen M, Dahlquist G, Persson B, Landin-Olsson M, Lernmark Å, Sundkvist G, Thalme B (1988) Factors influencing the magnitude, duration, and rate of fall of B-cell function in Type 1 (insulin-dependent) diabetic children followed for two years from their clinical diagnosis. Diabetologia 31: 664–669
Sochett EB, Daneman D, Clarson C, Ehrlich RM (1987) Factors affecting and patterns of residual insulin secretion during the first year of Type 1 (insulin-dependent) diabetes mellitus in children. Diabetologia 30: 453–459
Heding LG, Ludvigsson J, Kasperska-Czyzykowa T (1981) Bcell secretion in non-insulin and insulin-dependent-diabetics. Acta Med Scand [Suppl.] 659: 5–9
Ludvigsson J, Heding LG (1982) Abnormal proinsulin/C-peptide ratio in juvenile diabetes. Acta Diabet Lat 19: 351–358
The Canadian-European randomized control trial group (1988) Cyclosporin-induced remission of IDDM after early intervention: association of 1 yr of cyclosporin treatment with enhanced insulin secretion. Diabetes 37: 1574–1582
Feutren G, Papoz L, Assan R, Vialettes B, Karsenty G, Vexiau P, Du Rostu H, Rodier M, Sirmai J, Lallemand A, Bach JF (1986) Cyclosporin increases the rate and length of remission in insulin dependent diabetes of recent onset. Lancet II: 119–123
Heding LG (1975) Radioimmunological determination of human C-peptide in serum. Diabetologia 11: 541–548
Faber OK, Markussen J, Naithani VK, Binder C (1976) Production of anitsera to synthetic benzyloxycarbonyl-C-peptide of human proinsulin. Hoppe Seylers Z Physiol Chem 357: 751–757
Hartling SG, Dinesen B, Kappelgård A-M, Faber OK, Binder C (1986) ELISA for human proinsulin. Clin Chim Acta 156: 289–297
The Canadian/European Diabetes Study Group. Prepared by: Mandrup-Poulsen T, Mølvig J, Andersen HU, Helquist S, Munck M (1990) Lack of predictive value of islet cell antibodies, insulin autoantibodies, and HLA-DR phenptype for remission in Type I (insulin-dependent) diabetic patients treated with cyclosporin. Diabetes (in press)
Gordon P, Hendricks CM, Roth J (1974) Circulating proinsulin-like component in man: increased proportions in hypoinsulinemic states. Diabetologia 10: 469–474
Hartling SG, Lindgren F, Dahlqvist G, Persson B, Binder C (1989) Proinsulin is elevated in healthy siblings of insulin dependent diabetics and independent of HLA identity. Diabetes 38: 1271–1274
Glaser B, Leibovich G, Nesher R, Hartling SG, Binder C, Cerasi E (1988) Improved beta-cell function after intensive insulin treatment in severe non-insulin-dependent diabetes. Acta Endocrinol 118: 365–373
Yoshioka N, Kuzuya T, Matsuda A, Taniguchi M, Iwamoto Y (1988) Serum proinsulin levels at fasting and after oral glucose load in patients with Type 2 (non-insulin-dependent) diabetes mellitus. Diabetologia 31: 355–360
Yki-Järvinen H, Koivisto VA (1986) Natural course of insulin resistance in type I diabetes. N Eng J Med 316: 224–230
Koivisto VA, Yki-Järvinen H, Hartling SG, Pelkonen R (1986) The effect of exogenous hyperinsulinemia on proinsulin secretion in normal man. Obese subjects and patients with insulinoma. J Clin Endocrinol Metab 63: 1117–1120
Bergenstal RM, Cohen RM, Lever E, Polonsky K, Jaspan J, Blix PM, Revers R, Olefsky JM, Kolterman O, Steiner K, Cherrington A, Frank B, Galloway J, Rubenstein AH (1984) The metabolic effects of biosynthetic human proinsulin in individuals with type I diabetes. J Clin Endocrinol Metab 58: 973–979
Revers R, Henry R, Schmeiser L, Kolterman O, Cohen R, Bergenstal R, Polonsky K, Jaspan J, Rubenstein A, Frank B, Galloway J, Olefsky JM (1984) The effects of biosynthetic human proinsulin on carbohydrate metabolism. Diabetes 33: 762–770
Polonsky K, Licinio-Paixao J, Given BD, Pugh W, Rue P, Galloway J, Karrison T, Frank B (1986) Use of biosynthetic human C- peptide in the measurement of insulin secretion rates in normal volunteers and Type I diabetic patients. J Clin Invest 77: 98–105
Heding LG, Persson B, Stangenberg M (1980) B-cell function in newborn infants of diabetic mothers. Diabetologia 19: 427–432
Nerup J, Mandrup-Poulsen T, Mølvig J, Helqvist S, Wogensen L, Egeberg J (1988) Mechanisms of pancreatic β-cell destruction in type I diabetes. Diabetes Care 11 [Suppl. 1]: 16–23
McDaniel M, Hughes JH, Wolf BA, Easom RA, Turk JW (1988) Descriptive and mechanistic considerations of interleukin 1 and insulin secretion. Diabetes 37: 1311–1315
Rey AD, Besedovsky H (1987) Interleukin 1 affects glucose homeostasis. Am J Physiol 253: R794-R798
Jaspan JB, Mako ME, Kuzuya H, Blix PM, Horwitz DL, Rubinstein AH (1977) Abnormalities in circulating beta cell peptides in chronic renal failure: comparison of C-peptide, proinsulin and insulin. J Clin Endocrinol Metab 45: 441–446
Author information
Authors and Affiliations
Additional information
Prepared by the authors for The Canadian/European Diabetes Study Group.
Rights and permissions
About this article
Cite this article
Snorgaard, O., Hartling, S.G. & Binder, C. Proinsulin and C-peptide at onset and during 12 months cyclosporin treatment of Type 1 (insulin-dependent) diabetes mellitus. Diabetologia 33, 36–42 (1990). https://doi.org/10.1007/BF00586459
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00586459