Abstract
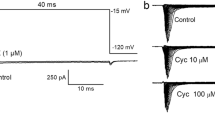
1. The effects of three toxins (ATX I, II, III) isolated from the sea anemoneAnemonia sulcata were studied in the soma membrane of a crustacean neurone under voltage-clamp conditions. 2. All three toxins affected the action potentials and the Na+ currents in a similar manner. The lowest concentrations tested (10 nM, 20 nM and 50 nM for AtX I, II and III, respectively) had pronounced selective effects on the Na+ current. No effect on K+ or Ca2+ currents was observed with concentrations up to 5 μM. 3. In the presence of ATX the Na+ inactivation was incomplete even with pulses of 700 ms length or strong depolarizing prepulses. 4. Besides the effects on the inactivation process ATX affected also the activation of the Na+ current. 5. In cells treated with ATX the negative resistance branch of the peak Na+ current voltage relation was shifted by −5 mV to −20 mV. 6. The time to peak was increased for small depolarizations (up to −30 mV) and the rate of rise (ΔI/Δt) was enlarged by ATX. A slow activating current component was also observed after depolarizing prepulses or if the Na+ current was outward. 7. The decay of the Na+ tail currents was considerably prolonged after the application of ATX if the membrane was repolarized to potentials more positive than about −60 mV. 8. Repetitive stimulation led to a shortening of the action potential in ATX II treated neurones. A simultaneous and parallel decrement of the peak and plateau current was observed with depolarizing voltage steps.
Similar content being viewed by others
References
Aldrich RW, Corey DP, Stevens CF (1983) A reinterpretation of mammalian sodium channel gating based on single channel recording. Nature 306:436–441
Alsen C (1983) Biological significance of peptides fromAnemonia sulcata. Fed Proc 42:101–108
Beress L (1982) Biologically active compounds from Coelenterates. Pure Appl Chem 54:1981–1994
Beress L, Ritter R, Ravens U (1982) The influence of the rate of electrical stimulation on the effects of theAnemonia sulcata toxin ATX II in guinea pig papillary muscle. Eur J Pharmac 79:265–272
Bergman C, Dubois JM, Rojas E, Rathmayer W (1976) Decreased rate of sodium conductance inactivation in the node of Ranvier induced by a polypeptide toxin from sea anemone. Biochim Biophys Acta 455:173–184
Erxleben C, Rathmayer W (1984) Effects ofAnemonia sulcata toxin II on neuromuscular transmission and skeletal muscle. Toxicon 22:387–399
Gillespie JI, Meves H (1980) The effect of scorpion venoms on the sodium currents of the squid giant axon. J Physiol 308:479–499
Hartung K, Rathmayer W (1983) Effects of three anemone toxins on sodium currents in a crayfish neurone. Naunyn-Schmiedeberg's Arch Pharmacol 322:R66
Hashimoto K, Ochi R, Hashimoto K, Inui J, Miura Y (1980) The ionic mechanism of prolongation of action potential duration of cardiac ventricular muscle by anthopleurin-A and its relationship to the inotropic effect. J Pharm Exp Ther 215:479–485
Low PA, Wu CH, Narahashi T (1979) The effect of anthopleurin-A on crayfish giant axon. J Pharm Exp Ther 210:417–421
Lund EA, Narahashi T (1981) Kinetics of sodium channel modification by the insecticide tetramethrin in squid axon membranes. J Pharm Exp Ther 219:464–473
Meves H, Rubly N, Watt DD (1982) Effect of toxins isolated from the venom of the scorpionCentruroides sculpturatus on the Na currents of the node of Ranvier. Pflügers Arch 393:56–62
Narahashi T, Moore JW, Shapiro BI (1969)Condylactis toxin: Interaction with nerve membrane ionic conductances. Science 163:680–681
Neumcke G, Schwarz W, Stämpfli R (1980) Modification of sodium inactivation in myelinated nerve byAnemonia toxin II and iodate: analysis of current fluctuations and current relaxations. Biochim Biophys Acta 600:456–466
Otsuka M, Kravitz EA, Potter DD (1967) Physiological and chemical architecture of a lobster ganglion with particular reference to gamma-amino butyrate and glutamate. J Neurophysiol 30:725–752
Patlak J, Horn R (1982) Effect of N-bromoacetamid on single sodium channel currents in excised membrane patches. J Gen Physiol 79:333–351
Pelhate M, Hue B, Satelle DB (1979) Pharmacological properties of axonal sodium channels in the cockroachPeriplaneta americana L. II. Slowing of sodium-current turn-off byCondylactis toxin. J Exp Biol 83:49–58
Rathmayer W (1979) Sea anemone toxins: tools in the study of excitable membranes. Adv Cytopharmacol 3:335–344
Rathmayer W, Beress L (1976) The effect of toxins fromAnemonia sulcata (Coelenterata) on neuromuscular transmission and nerve action potentials in the crayfish (Astacus leptodactylus). J Comp Physiol 109:373–382
Romey G, Abita JP, Schweitz H, Wunderer G, Lazdunski M (1976) Sea anemone toxin: A tool to study molecular mechanisms of nerve conduction and excitation-secretion coupling. Proc Natl Acad Sci USA 73:4055–4059
Rudy B (1981) Slow inactivation of voltage-dependent channels. In: Matsumoto G, Kotoni M (eds) Nerve membrane: Biochemistry and function of channel proteins. University of Tokyo Press, Tokyo, pp 89–111
Scheffler JJ, Tsugita A, Linden G, Schweitz H, Lazdunski M (1982) The amino acid sequence of toxin V fromAnemonia sulcata. Biochem Biophys Res Comm 107:272–278
Schmidtmayer J, Stoye-Herzog M, Ulbricht W (1982) Rate of action ofAnemonia sulcata toxin II on sodium-channels in myelinated nerve fibres. Pflügers Arch 394:313–319
Takeda K, Kennedy DJ (1964) Soma potentials and modes of activation of crayfish motoneurones. J Cell Comp Physiol 64:165–181
Ulbricht W, Schmidtmayer J (1981) Modification of sodium channels in myelinated nerve byAnemonia sulcata toxin II. J Physiol (Paris) 77:1103–1111
Warashina A, Fujita S (1983) Effect of sea anemone toxins on the sodium inactivation process in crayfish axons. J Gen Physiol 81:305–323
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hartung, K., Rathmayer, W. Anemonia sulcata toxins modify activation and inactivation of Na+ currents in a crayfish neurone. Pflugers Arch. 404, 119–125 (1985). https://doi.org/10.1007/BF00585406
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00585406