Abstract
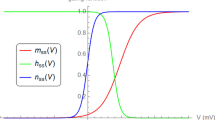
(1) Action potentials and membrane currents were recorded in single myelinated rat nerve fibres at 20 and 37° C. Three experiments were also performed in single cat nerve fibres. (2) K currents were blocked by internal CsCl and external TEA. The steady state and kinetic parameters of Na activation and inactivation were determined at both temperatures. (3) When the temperature was raised from 20 to 37° C, steady state Na activation,m ∞(V), and inactivation,h ∞(V), did not change significantly. (4) The time constant of Na activation, τm, was determined within the potential range of −40 to 125 mV at 20° C andV=40–60 mV at 37° C. The temperature coefficient, Q10, of τm was 2.2. (5) The decay in the Na current was described by two exponentials at both temperatures. The amplitude of the slow phase was 1–10%. The time constant of the fast phase of Na inactivation, τh1, was determined at both temperatures within the potential range of −50 mV to 125 mV. The Q10 of τh1 was 2.9 and did not depend on potential. (6) The Na equilibrium potential was 152 mV at 20° C and 144 mV at 37° C. The leakage conductance was 24 nS at 20° C and 43 nS at 37° C. These differences were interpreted as signs of fibre deterioration at higher temperature. (7) The results from the current and voltage clamp experiments performed in the cat nerve were essentially the same as those in the rat nerve fibres. (8) The action potentials computed on the basis of the voltage clamp results at 20° C were similar to the ones actually measured. This was also true for those action potentials predicted for 37° C on the basis of the 20° C data, theg L andV Na values measured at 37° C, and the Q10 values of the time constants. (9) Steady state values and kinetic parameters of K permeability were adopted from the literature. As in the experiments the influence ofP K on the shape of the predicted action potential was almost negligible at both temperatures.
Similar content being viewed by others
References
Bostock H, Sears TA, Sherratt RM (1981) The effects of 4-aminopyridine and tetraethylammonium ions on normal and demyelinated mammalian nerve fibres. J Physiol 313:301–315
Brismar T (1980) Potential clamp analysis of membrane currents in rat myelinated nerve fibres. J Physiol 298:171–184
Brismar T, Schwarz JR (1985) Potassium permeability in rat myelinated nerve fibres. Acta Physiol Scand 124:141–148
Bromm B, Frankenhaeuser B (1972) Repetitive discharge of the excitable membrane computed on the basis of voltage clamp data for the node of Ranvier. Pflügers Arch 332:21–27
Bromm B, Schwarz JR, Ochs G (1981) A quantitative description of combined potential and current clamp experiments on the single myclinated nerve fibre ofRana esculenta. J Theor Neurobiol 1:120–133
Chiu SY (1977) Inactivation of sodium channels: second order kinetics in myelinated nerve. J Physiol 273:573–596
Chiu SY (1980) Asymmetry currents in mammalian myelinated nerve. J Physiol 309:499–519
Chiu SY, Ritchie JM (1981) Evidence for the presence of potassium channels in the internodal region of acutely demyelinated mammalian single nerve fibres. J Physiol 313:415–437
Chiu SY, Mrose HE, Ritchie JM (1979a) Anomalous temperature dependence of sodium conductance in rabbit nerve compared with frog nerve. Nature 279:327–328
Chiu SY, Ritchie JM, Rogart RB, Stagg D (1979b) A quantitive description of membrane currents in rabbit myelinated nerve. J Physiol 292:149–166
Dronin H, Neumcke B (1974) Specific and unspecific charges at the sodium channel of the nerve membrane. Pflügers Arch 351:207–229
Frankenhaeuser B (1960) Sodium permeability in toad nerve and in squid nervel. J Physiol 152:159–166
Frankenhaeuser B, Huxley AF (1964) The action potential in the myelinated nerve fibre ofXenopus laevis as computed on the basis of voltage clamp data. J Physiol 171:302–315
Frankenhaeuser B, Moore LE (1963) The effect of temperature on sodium and potassium permeability changes in myelinated nerve fibres ofXenopus laevis. J Physiol 169:431–437
Hille B (1977) Ionic basis of resting and action potentials. In: Brookhart JM, Mountcastle VB, Kandel ER, Geiger SR (eds) Handbook of physiology. The nervous system I. American Physiological Society. Washington, DC, pp 99–136
Hodgkin AL, Huxley AF (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol 117:500–544
Horákova M, Nonner W, Stämpfli R (1968) Action potentials and voltage clamp currents of single rat Ranvier nodes. Proc Int Union Physiol Sci 7:198
Huxley AF (1959) Ion movements during nerve activity. Ann NY Acad Sci 81:221–246
Kniffki K-D, Siemen D, Vogel W (1981) Development of sodium permeability inactivation in nodal membranes. J Physiol 313:37–48
Meves H, Rubly N (1986) Kinetics of sodium current and gating current in the frog node of Ranvier. Pflügers Arch 407:18–26
Moore LE (1971) The effect of temperature and calcium ions on the rate constants of myelinated nerve. Am J Physiol 221:131–137
Neumcke B, Stämpfli R (1982) Sodium currents and sodium-dependent fluctuations in rat myelinated nerve fibres. J Physiol 329:163–284
Neumcke B, Schwarz JR, Stämpfli R (1987) A comparison of sodium currents in rat and frog myelinated nerve: normal and modified sodium inactivation. J Physiol 382:175–191
Noda M, Shimizu S, Tanabe T, Takai T, Kayano T, Ikeda T, Takahashi H, Nakayama H, Kanaoka Y, Minamina N, Kangawa K, Matsuo H, Raftery MA, Hirose T, Iniyama S, Hayashida H, Miyata T, Numa S (1984) Primary structure ofElectrophorus electricus sodium channel deduced from cDNA sequence. Nature 312:121–192
Nonner W (1969) A new voltage clamp method for Ranvier nodes. Pflügers Arch 309:176–192
Nonner W, Stämpfli R (1969) A new voltage clamp method. In: Passow H, Stämpfli R (eds) Laboratory techniques in membrane biophysics. Springer, Berlin Heidelberg New York, pp 171–175
Ochs G, Bromm B, Schwarz, JR (1981) A three-state model for inactivation of sodium permeability. Biochim Biophys Acta 645:243–252
Rogart RB, Stämpfli R (1982) Voltage-clamp studies of mammalian myelinated nerve. In: Culp WJ, Ochoa J (eds) Abnormal nerves and muscles as impulse generators. Oxford University Press, New York Oxford, pp 193–201
Schmidt H, Stämpfli R (1964) Nachweis unterschiedlicher elektrophysiologischer Eigenschaften motorischer und sensibler Nervenfasern des Frosches. Helv Physiol Pharmacol Acta 22:C143-C145
Schmidt H, Stämpfli R (1966) Die Wirkung von Tetraethylammoniumchlorid auf den einzelnen Ranvierschen Schnürring. Pflügers Arch 287:311–325
Schwarz JR (1986) The effect of temperature on Na currents in rat myelinated nerve fibres. Pflügers Arch 406:397–404
Schwarz JR (1987) Action potentials and repetitive activity in rat nerve: measurements and predictions. Pflügers Arch 408:R276
Schwarz JR, Eikhof G (1987) Membrane currents in rat myelinated nerve fibres at 20 and 37° C. Pflügers Arch 408:R264
Schwarz JR, Bromm B, Spielmann RP, Weytjens JLF (1983) Development of Na inactivation in motor and sensory myelinated nerve fibres ofRana esculenta. Pflügers Arch 398:126–129
Schwarz W (1979) Temperature experiments on nerve and muscle membranes of frogs. Indications for a phase transition. Pflügers Arch 382:27–34
Spielmann RP, Schwarz JR, Bromm B (1983) Oszillating repolarization in action potentials of frog sensory myelinated nerve fibres. Neurosci Lett 36:49–53
Stämpfli R, Hille B (1976) Electrophysiology of the peripheral nerve. In: Llinás R, Precht W (eds) Handbook of frog neurobiology. Springer, Berlin Heidelberg New York, pp 3–32
Vierhaus J, Ulbricht W (1969) Rate of action of tetraethylammonium ions on the duration of action potentials in single Ranvier nodes. Pflügers Arch 326:88–100
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwarz, J.R., Eikhof, G. Na currents and action potentials in rat myelinated nerve fibres at 20 and 37° C. Pflugers Arch. 409, 569–577 (1987). https://doi.org/10.1007/BF00584655
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00584655