Summary
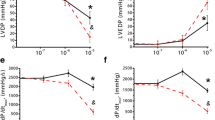
Infusion of adenosine into the coronary arteries of isolated guinea pig hearts produced a dosedependent inhibition of dP/dtmax caused by bolus injections of isoproterenol (4×10−11 moles). Threshold concentration of adenosine was 10−7 M and maximal inhibition (90%) occurred at 10−5 M. Coronary dilation induced by, papaverine did not influence the contractile response to catecholamines. In addition to its influence on cardiac performance, adenosine (10−5 M) effectively inhibited the isoproterenol (10−7 M) induced initial rise in myocardial levels of cyclic 3′5′-AMP, glucose-1-phosphate and glucose-6-phosphate. Adenosine also antagonized the effect of isoproterenol on adenylate cyclase activity in a crude membrane preparation from guinea pig ventricles; it was without effect on the activity of the membrane phosphodiesterase. Theophylline inhibited the actions of adenosine both on adenylate cyclase activity and on contractile force development.-Upon infusion of isoproterenol (3×10−7 M) into the coronary arteries of the isolated heart (perfusion at constant pressure), the adenosine concentration in the effluent perfusate increased within 45 s from 10−8 M to about 10−6 M. It thus appears conceivable that in ventricular myocardium endogenously formed adenosine may serve 2 functions: dilation of the coronary arteries and limitation of the inotropic and metabolic effects of catecholamines.
Similar content being viewed by others
References
Afonso, S.: Inhibition of coronary vasodilating action of dipyridamole and adenosine by aminophylline. Circ. Res.26, 743–752 (1970)
Arnold, G., Kosche, F., Miessner, E., Neitzert, A., Lochner, W.: The importance of perfusion pressure in the coronary arteries for the contractility and oxygen consumption of the heart. Pflügers Arch.299, 339–356 (1968)
Bergmeyer, H. U., Michal, G.: D-glucose-1-phosphate. In: Methoden der enzymatischen Analyse (H. U. Bergmeyer, ed.), pp. 1278–1282. Weinheim: Verlag Chemie 1974
Berne, R. M.: Cardiac nucleotides in hypoxia: possible role in regulation of coronary blood flow. Am. J. Physiol.204, 317–322 (1963)
Buckley, N. M.: Cardiac effects of nucleosides after propranolol treatment of isolated hearts. Am. J. Physiol.218, 1399–1405 (1970)
Bünger, R., Haddy, F. J.., Gerlach, E.: Coronary responses to dilating substances and competitive inhibition by theophylline in the isolated perfused guinea pig heart. Pflügers Arch.358, 213–224 (1975)
Cheung, W. Y., Williamson, J. R.: Kinetics of cyclic adenosine monophosphate changes in rat heart following epinephrine administration. Nature207, 979–981 (1970)
Drummond, G. I., Duncan, L.: Adenylate cyclase in cardiac tissues J. Biol. Chem.245, 976–983 (1970)
Drury, A., Szent-Gyögyi A.: The physiological activity of adenine compounds with special reference to their action upon mammalian heart. J. Physiol. (Lond.)68, 213–226 (1929)
Engelhard, V. H., Plut, D. A., Storm, D. R.: Subcellular location of adenylate cyclase in rat cardiac muscle. Biochim. Biophys. Acta451, 48–62 (1976)
De Gubareff, T., Sleator, W.: Effects of caffeine on mammalian atrial muscle and its interactions with adenosine and calcium. J. Pharmacol. Exp. Ther.148, 202–214 (1965)
Gilman, A. G.: A protein-binding assay for adenosine 3′5′-cyclic monophosphate. Proc. Natl. Acad. Sci. U.S.A.67, 303–331 (1970)
Fain, J. N., Pointer, R. H., Ward, W. F.: Effects of adenosine nucleosides on adenylate cyclase, phosphodiesterase, cyclic adenosine monophosphate accumulation, and lipolysis in fat cells. J. Biol. Chem.247, 6866–6872 (1972)
Lammerant, J., Becsei, I.: Left ventricular contractility and developed tension in the intact dog submitted to an intracoronary infusion of adenosine. J. Physiol. (Lond.)229, 41–49 (1973)
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with the Folin phenol reagent. J. Biol. Chem.193, 265–275 (1951)
McKenzie, S. G., Bär, H. P.: On the mechanism of adenyl cyclase inhibition by adenosine. Can. J. Physiol. Pharmacol.51, 190–196 (1973)
Rodbell, M., Londos, C.: Regulation of hepatic adenylate cyclase by glucagon, GTP, divalent cations and adenosine. Metabolism25, 1347–1351 (1976)
Rubio, R., Berne, R. M., Dobson, J. G.: Sites of adenosine production in cardiac and skeletal muscle. Am. J. Physiol.225, 783–800 (1973)
Sattin, A., Rall, T. W.: The effect of adenosine and adenine nucleotides on the cyclic adenosine 3′5′-phosphate content of guinea pig cerebral cortex slices. Mol. Pharmacol.6, 13–23 (1970)
Schrader, J., Rubio, R., Berne, R. M.: Inhibition of slow action potentials of guinea pig atrial muscle by adenosine: a possible effect, on Ca2+-influx. J. Mol. Cell. Cardiol.7, 427–433 (1975)
Schrader, J., Gerlach, E.: Compartmentation of cardiac adenine nucleotides and formation of adenosine. Pflügers Arch.367, 129–135 (1976)
Schwabe, U., Schönhofer, P. S., Ebert, R.: Facilitation by adenosine of the action of insulin on the accumulation of adenosine 3′5′-monophosphate, lipolysis, and glucose oxidation in isolated fat cells. Eur. J. Biochem.46, 537–545 (1974)
Shimizu, H., Daly, J. W., Creveling, C. R.: A radioisotopic method for measuring the formation of adenosine 3′,5′-cyclic monophosphate in incubated slices of brain. J. Neurochem.16, 1609–1619 (1969)
Shah, A., Kechejian, S. J., Kavaler, F., Fisher, V. J.: Effects of adenine nucleotides on contractility of normal and postischemic myocardium. Am. Heart J.87, 740–749 (1974)
Sonnenschein, r. R., Wright, D. L., Mellander, S.: Effects of vasodilators on filtration coefficient and distal arterial pressure in muscle. Am. J. Physiol.213, 706–710 (1967)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schrader, J., Baumann, G. & Gerlach, E. Adenosine as inhibitor of myocardial effects of catecholamines. Pflugers Arch. 372, 29–35 (1977). https://doi.org/10.1007/BF00582203
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00582203