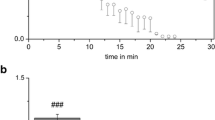
Abstract
Transitional cells of the crista ampullaris were impaled with microelectrodes in order to record the membrane potential (PD) and to investigate membrane properties. In control solution the PD was −87±1 mV (n=103). This value is not significantly different from −83±2 mV (n=24) measured in Cl− free solution. [Cl−] steps from 150 to 15 mmol/l (n=24) depolarized the membrane by about 2 mV, indicating a minor Cl− conductance. The transference number for K+ was 0.75±0.01 (n=79) obtained from the PD responses to K+ steps from 3.6 to 25 mmol/l. The cell membrane depolarized and the amplitude of PD responses to [K+] steps was reduced by Ba2+ (2·10−6 to 10−3 mol/l), quinidine (10−3 mol/l), quinine (10−3 mol/l), Rb+ (20 mmol/l), Cs+ (20 mmol/l), NH4 + (20 mmol/l) and Tl+ (0.5 mmol/l), whereas tetraethylammonium (TEA, 20 mmol/l) had no effect. The dose-response curve for Ba2+ in the presence of 3.6 mmol/l K+ was shifted to the right by approximately three decades in the presence of 25 mmol/l K+ and by a factor of about 4 in the presence of 135 mmol/l gluconate as a substitute for Cl−. Transitional cells were depolarized by ouabain, suggesting the presence of (Na++K+-ATPase.
Similar content being viewed by others
References
Adelman WJ, French RJ (1978) Blocking of the squid axon potassium channel by external cesium ions. J Physiol 276:13–25
Adrian RH, Chandler WK, Hodgkin AL (1970) Slow changes in potassium permeability in skeletal muscle. J Physiol (Lond) 208:645–668
Bear ChE, Davison JS, Shaffer EA (1988) Intracellular pH influences resting membrane potential of isolated rat hepatocytes. Biochim Biophys Acta 944:113–120
Bezanilla F, Armstrong CM (1972) Negative conductance caused by entry of sodium and cesium ions into the potassium channels of the squid axons. J Gen Physiol 60:588–608
Boron WF, De Weer P (1979) Intracellular pH transients in squid giant axons caused by CO2, NH3, and metabolic inhibitors. J Gen Physiol 67:91–112
Burckhardt B-Ch, Cassola AC, Frömter E (1984) Electrophysiological analysis of bicarbonate permeation across the peritubular cell membrane of rat kidney proximal tubule. II. Exclusion of HCO3 −-effects on other ion permeabilities and of coupled electroneutral HCO3 −-transport. Pflügers Arch 401:43–51
Demarest JR, Finn AL (1987) characterization of the basolateral membrane conductance of Necturus urinary bladder. J Gen Physiol 89:541–562
Findlay I, Dunne MJ, Ullrich J, Wollheim CB, Petersen OH (1985) Quinine inhibits Ca++-independent K+ channels whereas tetraethylammonium inhibits Ca++-activated K+ channels in insulin-secteting cells. FEBS Lett 185:4–8
Frindt G, Palmer LG (1987) Ca-activated K channels in apical membrane of mammalian CCT, and their role in K+ secretion. Am J Physiol 252:F458-F467
Gallacher DV, Maruyama Y, Petersen OH (1984) Patch-clamp study of rubidium and potassium conductances in single cation channels from mammalian exocrine acini. Pflügers Arch 401:361–367
Gay LA, Stanfield PR (1977) Cs+ causes a voltage dependent block on inward K+ currents in resting skeletal muscle fibres. Nature (Lond) 267:169–170
Gögelein H, Greger R, Schlatter E (1987) Potassium channels in the basolateral membrane of the rectal gland ofSqualus acanthias. Regulation and inhibitors. Pflügers Arch 409:107–113
Greger R, Gögelein H (1987) Role of K+ conductive pathway in the nephron. Kidney Int 31:1055–1064
Guggino SE, Guggino WB, Green N, Sacktor B (1987) Blocking agents of Ca++-activated K+-channels in cultured medullary thick ascending limb cells. Am J Physiol 252:C128-C137
Hagiwara S, Takahashi K (1974) The anomalous rectification and cation selectivity of the membrane of starfish egg cell. J Membr Biol 18:61–80
Hagiwara S, Miyazaki S, Rosenthal NP (1976) Potassium current and the effect of cesium on this current during anomalous rectification of the egg cell membrane of a starfish. J Gen Physiol 67:621–638
Lorenzen M, Lee CO, Windhager EE (1984) Cytosolic Ca++ and Na+ activities in perfused proximal tubules of Necturus kidney. Am J Physiol 247:F93-F102
Marcus DC, Wangemann P, Greger R (1988) Intracellular membrane potentials of epithelial cells in the ampulla of the Gerbil. FASEB J 2:A753
Messner G, Wang W, Paulmichl M, Oberleitner H, Lang F (1985) Ouabain decreases apparent potassium-conductance in proximal tubules of the amphibian kidney. Pflügers Arch 404:131–137
Paulmichl M, Gstraunthaler G, Lang F (1985) Electrical properties of Madin-Darby canine kidney cells. Effects of extracellular potassium and bicarbonate. Pflügers Arch 405:102–107
Petersen OH, Findlay I (1987) Electrophysiology of the pancreas. Physiol Rev 67:1054–1116
Richards NW, Dawson DC (1986) Single potassium channels blocked by lidocaine and quinidine in isolated turtle colon epithelial cells. Am J Physiol 251:C85-C89
Stanfield PR (1987) Nucleotides such as ATP may control the activity of ion channels. TINS 10:335–339
Van Driessche W, Hillyard SD (1985) Quinidine blockage of K+ channels in the basolateral membrane of larval bullfrog skin. Pflügers Arch 405 (Suppl 1):S77-S82
Van Driessche W, Zeiske W (1980) Spontaneous fluctuations of potassium channels in the apical membrane of frog skin. J Physiol 299:101–116
Vergara C, Latorre R (1983) Kinetics of Ca++-activated K+ channels from rabbit muscle incorporated into planar bilayers. Evidence for Ca++ and Ba++ blockade. J Gen Physiol 82: 543–568
Völkl H, Geibel J, Lang F (1987) Effect of temperature on peritubular cell membrane potential and potassium conductance in isolated perfused proximal tubules of the mouse. Bioelectrochemistry and Bioenergetics 17:419–427
Wangemann Ph, Marcus D (1989) Membrane properties of transitional cells of the Gerbil ampulla: K+ conductance, Na+/H+ exchanger, (Na++K+)-ATPase. FASEB J 3:A564
Zeiske W, Van Driessche W (1979) Saturable K+ pathway across the outer border of frog skin (Rana temporaria): Kinetics and inhibition by Cs+ and other cations. J Membr Biol 47:77–96
Author information
Authors and Affiliations
Additional information
This work was supported by grants from the Deafness Research Foundation to PhW and the National Institute of Health (NS 19490) to DCM
Rights and permissions
About this article
Cite this article
Wangemann, P., Marcus, D.C. Membrane potential measurements of transitional cells from the crista ampullaris of the Gerbil. Pflugers Arch. 414, 656–662 (1989). https://doi.org/10.1007/BF00582132
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00582132