Abstract
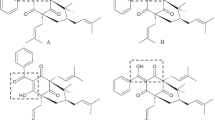
The results are given of a study of the13C NMR spectra of the new terpenoid coumarin bungeidiol (I) and of the products of its transformation (II) and (III) and some model compounds (IV–VII). On the basis of the results obtained from these13C NMR spectra and with the use of additive contributions depending on the nature and positions of various substituents (hydroxy and methoxy groups) in the aromatic ring, the structure (I) has been confirmed and a complete assignment of the signals of all the carbon atoms both in the coumarin ring and in the aliphatic part of the molecule of (I) has been made.
Similar content being viewed by others
Literature cited
N. J. Cussans and T. N. Huckerby, Tetrahedron,31, 2715 (1975).
O. A. Subbotin, P. I. Zakharov, V. A. Zagorevskii, and D. A. Zykov, Khim. Prir. Soedin., 458 (1975).
A. Patra et al., Indian J. Chem.,17B, 385 (1979).
C. Chang, H. Floss, and W. Steck, J. Org. Chem.,42, 1337 (1977).
G. Levy and G. Nelson, Carbon-13 in Nuclear Magnetic Resonance for Organic Chemists, Wiley-Interscience, New York (1972).
R. Freeman and H. D. Hill, J. Magn. Res.,5, 278 (1971).
F. Bohlmann, R. Zeisber, and E. Klein, Org. Magn. Reson.,7, 426 (1975).
I. B. Stothers, Carbon-13 NMR Spectroscopy, Academic Press, New York (1972) p. 13.
Organic Reactions [Russian translation], No. 1 (1948), p. 267.
A. Ray et al., Indian J. Chem.,16B, 929 (1978).
A. A. Perkin and M. Nierenstein, J. Chem. Soc.,87, 1412 (1905).
Additional information
Institute of Toxicology, Ministry of Health of the USSR, Leningrad. Translated from Khimiya Prirodynkh Soedinenii, No. 3, pp. 294–301, May–June, 1982.
Rights and permissions
About this article
Cite this article
Abyshev, A.Z., Zmeikov, V.P. 13C NMR spectra and structure of bungeidiol and its transformation products. Chem Nat Compd 18, 270–276 (1982). https://doi.org/10.1007/BF00580449
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00580449