Abstract
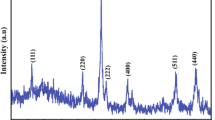
Seven ultrafine samples (125 to 60 m2/g) of the cobalt-iron oxide series, Co x Fe3−x ,O4, with 0≤x≤3, were prepared by co-precipitation of the metal hydroxides, followed by dehydration of the hydroxide at 100† C. X-ray and chemical analyses confirmed that true spinels were formed. The electrical conductivity and the activation energy for conduction of the powders were determined, and used to define the electronic structure of this series in conjunction with theoretical considerations based on crystal field theory.
Similar content being viewed by others
References
W. J. Schuele andV. D. Deetscreek, in “Ultrafine Particles”, ed. W. E. Kuhn (John Wiley, New York, 1963) p. 224.
G. Parravano, Fourth Int. Congress on Catalysis, Moscow 1968.
H. M. Cota, J. Katan, M. Chin, andF. J. Schoenweis,Nature 203 (1964) 1281.
A. F. Wells, “Structural Inorganic Chemistry” (Oxford University Press, 1962) p. 487.
J. P. Suchet, “Chemical Physics of Semiconductors” (Van Nostrand, Amsterdam, 1965) p. 77.
G. Blasse,Philips Res. Reports 18 (1963) 383.
G. H. Jonker,J. Phys. Chem. Solids 9 (1965) 165.
W. J. Schuele andV. D. Deetscreek,J. Appl. Phys. 32S (1961) 235.
A. C. C. Tseung, andH. L. Bevan,J. Mater. Sci. 5 (1970) 604.
T. Sato, M. Sugihara, andM. Saito,Rev. Elec. Commun. Lab. 11 (1963) 26.
R. S. Young, “Analytical Chemistry of Cobalt” (Pergamon Press, Oxford, 1965) p. 359.
J. Smiltens,J. Amer. Chem. Soc. 79 (1957) 4881.
T. M. Ovchinnikova,Doklady Acad. Nauk SSR 100 (1955) 469.
J. Besson, Ph.D. Thesis, University of Paris (1957).
N. V. Sidgwick, “The Chemical Elements and Their Compounds” (Oxford University Press, 1951) p. 1352.
C. L. Wilson andD. W. Wilson, Comprehensive Analytical Chemistry (Elsevier, Amsterdam, 1960) p. 170.
Y. A. Fialkov andN. V. Axelrud,Ukr. Khim. Zh. 16 (1950) 283.
W. C. Elmore,Phys. Rev. 54, (1938) 309.
J. R. Yates, B.Sc. Industrial Chemistry Project Report, The City University, London 1968.
W. R. Harper, “Electrical Properties of Powders” (Eyre and Spottiswoode, London, 1961) p. 117.
F. C. Romeijn,Philips Res. Reports 8 (1953) 304.
Gmelin, “Handbuch der Anorganischen Chemie” (Verlag Chemie, 1959) Vol. 59D, p. 369.
D. S. McClure,J. Phys. Chem. Solids 3 (1957) 311.
J. D. Dunitz andL. E. Orgel,ibid 3 (1957) 318.
J. Greenwald, R. Pickart, andF. Granis,J. Chem. Phys. 22 (1954) 1957.
E. Verwey,Philips Res. Reports 5 (1950) 173.
R. Bauminger andS. G. Cohen,Phys. Rev. 122 (1961) 743.
E. Prince,ibid 102 (1956) 674.
I. Dezsi,Acta Phys. Polon. 24 (1963) 283.
W. Müller andH. Schmalzried,Ber. Bunsengesell. Physik. Chem. 68 (1964) 270.
F. K. Lotgering,Philips Res. Reports 11 (1956) 337.
B. M. Hochberg andM. S. Sominski,Phys. Z. Sowjetunion 13 (1938) 198.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tseung, A.C.C., Goldstein, J.R. The preparation and characterization of ultrafine cobalt-iron oxides. J Mater Sci 7, 1383–1390 (1972). https://doi.org/10.1007/BF00574930
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00574930