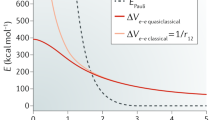
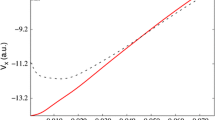
Abstract
Interaction energies for H2O·H2O, H2O·F− and H2O·CH4 have been calculated using the LCAO MO SCF method with minimal basis sets, and employing the counterpoise method to eliminate the basis set superposition error. The results compare favourably with those obtained using extended basis sets. It is shown that for H2O·H2O and for the benzene-carbonyl cyanide complex a large part of the dispersion energy can easily be obtained as a sum of bond-bond dispersion energies calculated from a London-type formula using experimental values of the bond polarizability tensors. By considering the interaction between a water and a glycine molecule it is also shown that the dispersion energy plays an important role in the hydration of organic molecules.
Similar content being viewed by others
References
Bertoncini, P., Wahl, A. C.: Phys. Rev. Letters25, 991 (1970)
McLaughlin, D. R., Schaefer, H. F.: Chem. Phys. Letters12, 244 (1972)
Liu, B., McLean, A. D.: J. Chem. Phys.59, 4557 (1973)
Diercksen, G. H. F., Kraemer, W. P., Roos, B. O.: Theoret. Chim. Acta (Berl.)36, 249 (1975)
Matsuoka, O., Clementi, E., Yoshimine, M.: J. Chem. Phys.64, 1351 (1976)
Jeziorski, B., van Hemert, M.: Mol. Phys.31, 713 (1976)
Jeziorski, B., Kołos, W.: Intern. J. Quantum Chem., in print
Jeziorski, B., Szalewicz, K., Chalasiński, G.: Intern. J. Quantum Chem., in print
Szalewicz, K., Jeziorski, B., to be published
Clementi, E., Cavallone, F., Scordamaglia, R.: J. Am. Chem. Soc.99, 5531 (1977)
Scordamaglia, R., Cavallone, F., Clementi, E.: J. Am. Chem. Soc.99, 5545 (1977)
Bolis, G., Clementi, E.: J. Am. Chem. Soc.99, 5550 (1977)
Clementi, E.: J. Chem. Phys.46, 3851 (1967)
Kestner, N. R.: J. Chem. Phys.48, 252 (1968)
Boys, S. F., Bernardi, F.: Mol. Phys.19, 553 (1970)
Meunier, A., Levy, B., Berthier, G.: Theoret. Chim. Acta (Berl.)29, 49 (1973)
Johansson, A., Kollman, P., Rothenberg, S.: Theoret. Chim. Acta (Berl.)29, 167 (1973)
Urban, N., Hobza, P.: Theoret. Chim. Acta (Berl.)36, 207, 215 (1975)
Jönsson, B., Karlström, G., Wennerström, H.: Chem. Phys. Letters30, 58 (1975)
Pullman, A., Berthod, H., Gresh, N.: Intern. J. Quantum Chem. Symp.10, 56 (1976)
Ostlund, N. S., Merrifield, D. L.: Chem. Phys. Letters39, 612 (1976)
Wormer, P. E. S., van der Avoird, A.: J. Chem. Phys.62, 3326 (1975)
Kocjan, D., Koller, J., Ažman, A.: J. Mol. Struct.34, 145 (1976)
Bulski, M., Chałasiński, G.: Theoret. Chim. Acta (Berl.)44, 399 (1977)
Groen, T. P., van Duijneveldt, F. B.: Theoret. Chim. Acta (Berl.), in print
Umeyama, H., Morokuma, K.: J. Am. Chem. Soc.99, 1316 (1977)
Clementi, E.: Lecture Notes in Chemistry, Vol. 2: Determination of liquid water structure. Berlin: Springer-Verlag 1976
Popkie, H., Kistenmacher, H., Clementi, E.: J. Chem. Phys.59, 1325 (1973)
Kistenmacher, H., Popkie, H., Clementi, E.: J. Chem. Phys.58, 5627 (1973)
Tosi, C., Scordamaglia, R., Clementi, E., Wertz, D. H., Scheraga, H. A.: to be published
Van Duijneveldt, F. B.: IBM Technical Report, RJ945, December 10, 1971
Corongiu, G., Clementi, E.: J. Chem. Phys., submitted for publication
Kołos, W., Leś, A.: Intern. J. Quantum Chem.6, 1101 (1972)
Dill, J. D., Allen, L. C., Topp, W. C., Pople, J. A.: J. Am. Chem. Soc.97, 7220 (1975)
Lathan, W. A., Pack, G. R., Morokuma, K.: J. Am. Chem. Soc.97, 6624 (1975)
London, F.: Z. Physik. Chemie (B)11, 222 (1930), Trans. Faraday Soc.33, 8 (1937)
Claverie, P.: “Elaboration of approximate formulas for the interactions between large molecules” in: Intermolecular interactions: from diatomics to biopolymers, B. Pullman, ed. New York: J. Wiley 1978
Landolt-Börnstein, Zahlenwerte und Funktionen, Vol. I/3, p. 513. J. Bartelet al., Eds. Berlin: Springer-Verlag 1951
Le Fevre, C. G., Le Fevre, R. J. W.: Rev. Pure Appl. Chem.5, 261 (1955)
Egan, J. T., Swissler, T. J., Rein, R.: Intern. J. Quantum Chem., Quantum Biol. Symp.1, 71 (1974)
Zeiss, G. D., Meath, W. J.: Mol. Phys.30, 161 (1975)
Dalgarno, A.: Advan. Chem. Phys.12, 143 (1967)
Ref. [38], p. 511
Romano, S., Clementi, E.: Gazz. Chim. It.108, 319, (1978)
Author information
Authors and Affiliations
Additional information
On leave from the Quantum Chemistry Laboratory, Institute of Basic Problems of Chemistry, University of Warsaw, Pasteura 1, 02-093 Warsaw, Poland
Rights and permissions
About this article
Cite this article
Kołos, W. Possible improvements of the interaction energy calculated using minimal basis sets. Theoret. Chim. Acta 51, 219–240 (1979). https://doi.org/10.1007/BF00572929
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00572929