Abstract
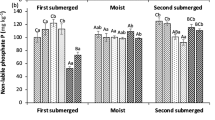
Simulated H2SO4 rain (pH 3.0, 3.5, 4.0) or control rain (pH 5.6) was applied for 3.5 yr to large lysimeter boxes containing a sulfate-adsorbing forest soil and either red alder (Alnus rubra Bong) or sugar maple (Acer saccharum Marsh.) seedlings. After removal of the plants and the litter layer, soil samples were obtained at 15-cm intervals to a total depth of 90 cm. Elevated SO4 concentrations caused by the simulated H2SO4 rain were most pronounced for the top 15 cm, but extended down to 45 cm (maple) or 75 cm (alder). There were no effects on SO4 concentrations at a depth of 75 to 90 em. This confirmed the existence of a sulfate front between 20 cm and 100 cm, as postulated earlier on the basis of extracted soil solutions. Decreases in Mg and Ca concentrations, base saturation, and soil pH were limited to the uppermost 15 cm and, in most cases, to the pH 3.0 treatment. Concentrations of Mg and Ca for the pH 3.0 treatments were greater than control at a depth of 15 to 30 cm, indicating transport of these cations from the soil surface. Concentrations of Na and K, and cation exchange capacity, were not affected by simulated H2SO4 rain. Elevated concentrations of NO3 and extractable Zn throughout the alder systems indicated (1) either increased rates of symbiotic N-fixation or decreased rates of N immobilization; and (2) mobilization of Zn by all acid rain treatments.
Similar content being viewed by others
References
Franklin, J. F., Dyrness, C. T., Moore, D. G., and Tarrant, R. F.: 1968, ‘Chemical Soil Properties Under Coastal Oregon Stands of Alder and Conifers’, in Trappe, J. M., Franklin, J. F., Tarrant, R. F., and Hansen, G. M. (eds.),Biology of Alder, Pacific Northwest Forest and Range Experiment Station, Forest Service, U.S. Department of Agriculture, Portland, OR. pp. 157–172.
Johnson, D. W. and Cole, D. W.: 1977, ‘Anion Mobility in Soils: Relevance to Transport from Terrestrial to Aquatic Ecosystems’, EPA 600/3-77-068, Environmental Protection Agency, Environmental Research Laboratory, Corvallis, OR.
Johnson, D. W. and Cole, D. W.: 1980,Environment International 3, 79.
Johnson, A. H. and Siccama, T. G.: 1983,Environ. Sci. Technol. 17, 294A.
Kaufman, M. D. and Gardner, E. H.: 1976, ‘Methods of Soil Analysis Used in the Soil Testing Laboratory at Oregon State University’, Special Report 321, Agricultural Experiment Station, Corvallis, OR.
Lee, J. J. and Weber, D. E.: 1982,J. Environ. Qual. 11, 57.
McFee, W. W.: 1980, ‘Sensitivity of Soil Regions to Acid Rain Precipitation’, EPA 600/3-80-013, Environmental Protection Agency, Environmental Research Laboratory, Corvallis, OR.
McFee, W. W., Adams, F., Cronan, C. S., Firestone, M. K., Foy, C. D., Harter, R. D., and Johnson, D. W.: 1984, ‘Effects on Soil Systems’, in Altshuller, A. P. and Linthurst, R. A. (eds.),The Acidic Deposition Phenomenon and its Effects: Critical Assessment Review Papers, EPA 600/8-83-016B, Environmental Protection Agency, Office of Research and Development, Washington, D.C. pp. E2.1–2.71.
Reuss, J. O.: 1980,Ecological Modeling 11, 15.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, J.J. Effect of simulated sulfuric acid rain on the chemistry of a sulfate-adsorbing forest soil. Water Air Soil Pollut 25, 185–193 (1985). https://doi.org/10.1007/BF00568387
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00568387