Summary
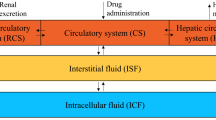
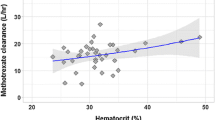
The pharmacokinetics of methotrexate have been assessed at two dose levels in six patients recciving the drug for treatment of malignant disease. Each patient received bolus intravenous doses of 25 mg and 100 mg given at least one week apart, the order of administration being random. Blood and urine were collected until 48 h for methotrexate analysis by radioimmunoassay and data analysed by a model-independent pharmacokinetic approach. In each patient area under the methotrexate serum concentration-time curve (o to ∞) increased out of proportion to the increase in methotrexate dose. This was reflected in a mean clearance value after the 100 mg dose of 31±16 (SD) ml · min−1 compared with a mean clearance of 62±19 ml · min−1 following injection of 25 mg methotrexate. Renal clearance of methotrexate was markedly lower following the 100 mg dose (18±6 ml · min−1) than after 25 mg (53±19 ml · min−1). Saturation of the proximal tubular organic acid transport system is the likely cause of methotrexate's capacity limited elimination.
Similar content being viewed by others
References
Calman KC, Lawrence JR, McVie JG, Sneader WE, Steele WH, Stuart JFB (1979) Methotrexate kinetics: effects of subdividing an oral dose. Br J Clin Pharmacol 7: 423–424
Chiou WL (1978) Critical evaluation of the potential error in pharmacokinetic studies of using the linear trapezoidal rule method for the calculation of area under the plasma level-time curve J Pharmacokinet Biopharm 6: 539–546
Goldie JH, Price LA, Harrap KR (1972) Methotrexate toxicity: Correlation with duration of administration, plasma levels, dose and excretion pattern. Eur J Cancer 8: 409–414
Huffman DH, Wan SK, Azarnoff DL, Hoogstraten B (1973) Pharmacokinetics of methotrexate. Clin Pharmacol Ther 14: 572–579
Leme PR, Creaven PJ, Allen LM, Berman M (1975) Kinetic model for the disposition and metabolism of moderate and high dose methotrexate (NSC — 740) in man. Cancer Chemother Rep 59: 811–817
Liegler DG, Henderson ES, Hahn MA, Oliverio VT (1969) The effect of organic acids on renal clearance of methotrexate in man. Clin Pharmacol Ther 10: 849–857
Paxton JW, Rowell FJ, Cree G (1978) Comparison of three radioligands75Selenium,125iodine and tritium in the radioimmunoassay of methotrexate. Clin Chem 24: 1534–1538
Pratt CB, Roberts D, Shanks EC, Warmathy EL (1974) Clinical trials and pharmacokinetics of intermittent high dose methotrexate “leucovorin resue” for children with malignant tumours. Cancer Res 34: 3326–3331
Reich DS, Bachur NR, Goebel RH, Bergman M (1977) A Pharmacokinetic model for high-dose methotrexate infusions in man. J Pharmacokinet Biopharm 5: 421–433
Shen DD, Azarnoff DL (1978) Clinical pharmacokinetics of methotrexate. Clin Pharmacokinet 3: 1–13
Stoller RG, Jacobs SA, Drake JC, Lutz RJ, Chabner BB (1975) Pharmacokinetics of high-dose methotrexate (NSC-740). Cancer Chemother Rep 6: 19–24
Wagner JG (1975) In: Fundamentals of clinical pharmacokineties. Drug Intelligence Publications, Hamilton, p 248
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lawrence, J.R., Steele, W.H., Stuart, J.F.B. et al. Dose dependent methotrexate elimination following bolus intravenous injection. Eur J Clin Pharmacol 17, 371–374 (1980). https://doi.org/10.1007/BF00558450
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00558450