Abstract
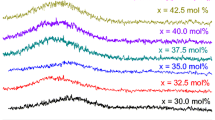
X-ray diffraction and infrared measurements were performed on vanadium borophosphate glass containing different amounts of iron ranging from 0–7.5 mol % and heat treated at 300 °C for various times. The structure and phase separation could be determined for each glass composition. V2O5 was the main precipitated phase in all heat-treated samples, and its amount was dependent on the heat-treatment time and Fe2O3 content. Also FeP was detected in samples heat treated for 24 h. The infrared measurements showed the presence of both V4+ and V5+. The symmetry of V2O 4−7 and VO 3−4 groups was found to increase with increasing Fe2O3 content. It was also found that some PO4 changed to BO3, forming a non-bridging oxygen.
Similar content being viewed by others
References
P. Baynton, H. Rawson andJ. Stanworth,Nature 178 (1956) 910.
G. W. Anderson andW. D. Compton,J. Chem. Phys. 53 (1970) 6166.
O. P. Hamblen, R. A. Weidel andG. E. Blair,J. Amer. Ceram. Soc. 46 (1963) 499.
Y. Limb andR. F. Davis,ibid. 62 (1979) 7.
B. Jordan andC. Calvo,Can. J. Chem. 51 (1973) 2621.
R. Gopal andC. Calvo,J. Solid State Chem. 5 (1972) 432.
R. H. Caley andM. Krishna Murthy,J. Amer. Ceram. Soc. 53 (1970) 254.
B. Nader,Class Ceram. 18 (1960) 174 (English Translation).
A. M. Abo-El-Azm, I. Kashif, H. Farouk, A. M. Sanad andY. M. Abo Zeid,Phys. Chem. Glasses 30(6) (1989) 12.
P. S. Rogers andJ. Williamson,J. Glass Technol. 10 (1969) 128.
P. W. McMillan, “Glass Ceramics”, 2nd Edn. (Academic Press, London, 1979).
A. Tomozawa,J. Amer. Ceram. Soc. 5 (1972) 41.
I. Sawal,Glass Technol. 2 (1961) 243.
I. R. Beattie andT. R. Gilson,J. Chem. Soc. A (1969) 2322.
E. J. Baron, I. L. Botle, J. F. Paous andP. J. Aymonins,ibid. 109 (1978) 1114.
Y. Dimitriev, V. Dimitrov, M. Arnaudov andD. Topalov,J. Non-Cryst. Solids 57 (1983) 147.
T. R. Gilson, O. F. Bizri andN. Cheetham,J. Chem. Soc. Dalton Trans. 3 (1973) 291.
G. Fabri andP. Baraldi,Anal. Chem. 44 (1972) 1325.
Ivan. Simon andH. O. McMohon,J. Chem. Phys. 20 (1952) 905.
B. C. Norman, H. D. Lawrence andE. W. Stephen, “Introduction to infrared spectroscopy” (Academic Press, New York, 1964).
H. G. Bachman, F. R. Ahmed andW. H. Barnes,Z. Kristallogr. 115 (1961) 110.
V. Sadagopan andH. C. Gatos,Mater. Sci. Engng 2 (1967) 273.
M. Irion andM. J. Couzie,J. Solid State Chem. 31 (1980) 285.
F. A. Moustafa, A. A. Abd El-Mongy, A. M. Sanad, I. Kashif andA. G. Moustafa,J. Mater. Sci. Lett. 4 (1985) 1054.
J. E. Stanworth,J. Amer. Ceram. Soc. 54 (1971) 61.
A. S. Tenney andJ. Wong,J. Chem. Phys. 56 (1972) 5516.
T. Abe,J. Amer. Ceram. Soc. 49 (1966) 11.
I. Kashif, A. M. Abo El-Azm, A. M. Sanad andE. E. Assem,J. Mater. Sci. Lett. 6 (1987) 1089.
I. Kashif, A. M. Sanad, A. A. El-Saghier, S. A. El-Sahhar andA. Abdel Razek,J. Mater. Sci. Lett. 5 (1986) 109.
Krogh-Moe,J. Phys. Chem. Glasses 6 (1965) 46.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kashif, I., Farouk, H., Sanad, A.M. et al. Structural studies of some V2O5-P2O5-B2O3-Fe2O3 glass systems. J Mater Sci 27, 122–126 (1992). https://doi.org/10.1007/BF00553846
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00553846