Abstract
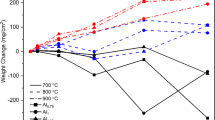
Three austenitic Fe-31 Mn-9Al-0.87C based alloys with various contents of chromium are oxidized in dry air from 800 to 1100° C. Decarburization takes place in the first stage of oxidation, and results in a porous initial alumina layer followed by a uniform and bulky oxide layer. In the second stage of oxidation or at higher temperature, the oxidation is dominated by the metallic elements. The chromium addition in this study acts as the oxygen getter to retard the oxidation of iron and especially decarburization in the first stage of oxidation. Qualitative and quantitative microscopic techniques are employed to investigate the morphological development and the elemental redistribution in the alloy system.
Similar content being viewed by others
References
P. R. S. Jackson and G. R. Wallwork, Oxid. Met. 21 (1984) 135.
J. P. Sauer, R. A. Rapp and J. P. Hirth, ibid. 18 (1982) 285.
R. Wang, M. J. Straszheim and R. A. Rapp, ibid. 21 (1984) 71.
J. G. Duh, C. J. Lin, J. W. Lee and C. M. Wan, “Alternate Alloying for Environmental Resistance”, edited by G. R. Smolik and S. K. Banerji (The Metallurgical Society, Warrendale, Pennsylvania, USA, 1987) p. 283.
J. G. Duh, C. J. Wang, C. M. Wan and B. S. Chiou, ibid“ p. 291.
C. J. Wang and J. G. Duh, J. Mater, Sci. 23 (1988) 769.
Idem. ibid. 23 (1988) 3447.
J. G. Duh, J. W. Lee and C. J. Wang, ibid 23 (1988) 2649.
P. Tomaszewicz and G. R. Wallwork, Oxid. Met. 20 (1983) 75.
G. B. Abderrazik, G. Moulin, A. M. Huntz and R. Berneron, J. Mater. Sci. 19 (1984) 3173.
L. J. Huetter and H. H. Stadelmaier, Acta. Metal. 6 (1958) 367.
E. R. Morral, J. Iron Steel Inst. 130 (1934) 419.
Power Diffraction File, JCPDS International Center for Diffraction Data, Swarthmore, Pennsylvania, 1979, file No. 29–44.
M. W. Chase, J. L. Curnutt, R. A. McDonald and A. N. Syverud, J. Phys. Chem. Ref. Data 7 (1978) 793.
C. B. Alcock and S. Zador, Electronchim. Acta. 12 (1967) 673.
Y. Jeannin, C. Mannerskantz and F. D. Richardson. Trans. Metall. Soc. AIME 227 (1963) 300.
D. R. Gaskell, “Introduction to Metallurgical Thermodynamics” (McGraw Hill, New York, 1983) p. 585.
D. R. Stull and H. Prophet, in “JANAF Thermochemical Tables”, NSRDS-NBS37, (US Department of Commerce, Washington, 1971).
W. C. Hahn and A. Muan, Amer. J. Sci. 258 (1960) 66.
E. M. Otto, J. Electronchem. Soc. 111 (1964) 88.
G. F. V. Voort, in “Metallography Principles and Practice” (McGraw Hill, New York, 1984) p. 426.
V. G. Rivlin, Internat. Met. Rev. 24 (1984) 299.
C. H. Kao, C. M. Wan and M. T. Jahn, in “Alternate Alloying for Environmental Resistance”, edited by G. R. Smolik and S. K. Banerji (The Metallurgical Society, Warrendale, Pennsylvania, USA, 1987) p. 299.
G. R. Smolik, J. E. Flinn, D. V. Miley and G. E. Korth, ibidin “ p. 307.
G. E. Hale and A. J. Baker, ibidin “ p. 67.
S. C. Chang, M. T. Jahn, Y. C. Pan and C. M. Wan in “Proceedings of the 7th International Conference on Strength of Metals and Alloy (Montreal, Canada, August, 1985).
A. U. Malik, Oxid. Met. 25 (1985) 233.