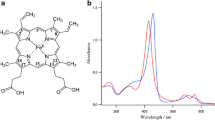
Abstract
The monomeric haemoglobin IV from Chironomus thummi thummi (CTT IV) exhibits an alkaline Bohr-effect and therefore it is an allosteric protein. By substitution of the haem iron for cobalt the O2 half-saturation pressure, measured at 25‡ C, increases 250-fold. The Bohr-effect is not affected by the replacement of the central atom. The parameters of the Bohr-effect of cobalt CTT IV for 25‡ C are: inflection point of the Bohr-effect curve at pH 7.1, number of Bohr protons -δlog p1/2 (O2)/gDpH=0.36 mol H+/mol O2 and amplitude of the Bohr-effect curve δlog p1/2 (O2)=0.84. The substitution of protoporphyrin for mesoporphyrin causes a 10 nm blue-shift of the visible absorption maxima in both, the native and the cobalt-substituted forms of CTT IV. Furthermore, the replacement of vinyl groups by ethyl groups at position 2 and 4 of the porphyrin system leads to an increase of O2 affinities at 25‡ C which follows the order: proto < meso < deutero for iron and cobalt CTT IV, respectively. Again, the Bohr-effect is not affected by the replacement of protoporphyrin for mesoporphyrin or deuteroporphyrin. The electron spin resonance (ESR) spectra of both, deoxy cobalt proto- and deoxy cobalt meso-CTT IV, are independent of pH. The stronger electron-withdrawing effect by protoporphyrin is reflected by the decrease of the cobalt hyperfine constants coinciding with g∥=2.035 and by the low-field shift of g∥. The ESR spectra of oxy cobalt proto- and oxy cobalt meso-CTT IV are dependent of pH. The cobalt hyperfine constants coinciding with g∥=2.078 increase during transition from low to high pH. The pH-induced ESR spectral changes correlate with the alkaline Bohr-effect. Therefore, the two O2 affinity states can be assigned to the low-pH and high-pH ESR spectral species. The low-pH form (low-affinity state) is characterized by a smaller, the high-pH form (high-affinity state) by a larger cobalt hyperfine constant in g∥. The correlation of the cobalt hyperfine constants of the oxy forms with the O2 affinities is discussed for several monomeric haemoglobins. The Co-O-O bond angle in cobalt oxy CTT IV is characterized by an ozonoid type of binding geometry and varies little during the pH-induced conformation transition. Due to the lack of the distal histidine in CTT IV no additional interaction via hydrogen-bonding with dioxygen is possible; this is reflected by the cobalt hyperfine constants.
Similar content being viewed by others
References
Gersonde K, Sick H, Wollmer A, Buse G (1972) Heterotropic allosterism of monomeric haemoglobins of Chironomus thummi thummi. Eur J Biochem 25: 181–189
Sick H, Gersonde K, Thompson JC, Maurer W, Haar W, Rüterjans H (1972) The Bohr proton-binding site in a monomeric haemoglobin. A nuclear-magnetic resonance study. Eur J Biochem 29: 217–223
Sick H, Gersonde K (1974) Ligand-specific Bohr-effect in haemoglobins. Eur J Biochem 45: 313–320
Gersonde K, Noll L, Gaud HT, Gill SJ (1976) A calorimetric study of the CO Bohr-effect of monomeric haemoglobins. Eur J Biochem 62: 577–582
Steffens G, Buse G, Wollmer A (1977) Ligand-dependent Bohr-effect of Chironomus hemoglobins. Eur J Biochem 72: 201–206
La Mar GN, Overkamp M, Sick H, Gersonde K (1978) Proton nuclear magnetic resonance hyperfine shifts as indicators of tertiary structural changes accompanying the Bohr-effect in monomeric insect hemoglobins. Biochemistry 17: 352–361
La Mar GN, Anderson RR, Budd DL, Smith KM, Langry KC, Gersonde K, Sick H (1981) Proton nuclear magnetic resonance investigation of the nature of the solution conformational equilibria of monomeric insect deoxy hemoglobins. Biochemistry 20: 4429–4436
Buse G, Steffens GJ, Braunitzer G, Steer W (1979) HÄmoglobin (Erythrocruorin) CTT III aus Chironomus thummi thummi (Diptera). PrimÄrstruktur und Beziehung zu anderen HÄmoglobinen. Hoppe-Seylers Z Physiol Chem 360: 89–97
Pfletschinger J, Plagens H, Braunitzer G (1980) The primary structure of the monomeric hemoglobin (Erythrocruorin) component CTT IV of Chironomus thummi thummi (Insecta, Diptera). Z Naturforsch 35c: 840–843
Steigemann W, Weber E (1979) Structure of Erythrocruorin in different ligand states refined at 1.4 å resolution. J Mol Biol 127: 309–338
Overkamp M, Twilfer H, Gersonde K (1976) Conformation-controlled trans-effect of the proximal histidine in haemoglobins. An electron spin resonance study of monomeric nitrosyl-57Fe-haemoglobins. Z Naturforsch 31c: 524–533
Twilfer H, Gersonde K (1976) Non-equivalence and inverse allosteric response of the α and Β chains in haemoglobins. An electron spin resonance study of NO-ligated Hb Kansas. Z Naturforsch 31c: 664–674
Huber R, Epp O, Formanek H (1970) Structures of deoxy and carbonmonoxy erythrocruorin. J Mol Biol 52: 349–354
Heidner EJ, Ladner RC, Perutz MF (1976) Structure of horse carbonmonoxyhaemoglobin. J Mol Biol 104: 707–722
Stryer L, Kendrew JC, Watson HC (1964) The mode of attachment of the azide ion to sperm whale metmyoglobin. J Mol Biol 8: 96–104
Perutz MF, Mathews FS (1966) An X-ray study of azide methaemoglobin. J Mol Biol 21: 199–202
Yonetani T, Yamamoto H, Iizuka T (1974) Studies on cobalt myoglobins and hemoglobins. III. Electron spin paramagnetic resonance studies of reversible oxygenation of cobalt myoglobins and hemoglobins. J Biol Chem 249: 2168–2174
Dickinson C, Chien JCW (1980) Electron paramagnetic resonance crystallography of 17O-enriched oxycobaltomyoglobin: Stereoelectronic structure of the cobalt dioxygen system. Proc Natl Acad Sci USA 77: 1235–1239
Asher SA, Adams ML, Schuster TM (1981) Resonance Raman and absorption spectroscopic detection of distal histidine-fluoride interactions in human methemoglobin fluoride and sperm whale metmyoglobin fluoride: Measurements of distal histidine ionization constants. Bio-chemistry 20: 3339–3346
Christahl M, Raap A, Gersonde K (1981) pH dependence of oxy and deoxy cobalt-substituted leghemoglobin from soybean. Biophys Struct Mech 7: 171–186
Griffith JS (1956) On the magnetic properties of some haemoglobin complexes. Proc R Soc (Lond) Ser A 235: 23–36
Pauling L (1964) Nature of the iron-oxygen bond in oxy haemoglobin. Nature 203: 182–183
Sugita Y, Yoneyama Y (1971) Oxygen equilibrium of hemoglobins containing unnatural hemes. Effect of modification of heme carboxyl groups and side chains at positions 2 and 4. J Biol Chem 246: 389–394
Seybert DW, Moffat K, Gibson QH (1976) Ligand binding properties of horse hemoglobins containing deutero- and mesoheme. J Biol Chem 251: 45–52
Fioretti E, Ascoli F, Brunori M (1976) Preparation of apohemoglobin trout IV and reconstitution with different hemes. Biochem Biophys Res Commun 68: 1169–1173
La Mar GN, Viscio DB, Gersonde K, Sick H (1978) Proton nuclear magnetic resonance study of the rotational position of oscillatory mobility of vinyl groups in allosteric monomeric insect hemoglobins. Biochemistry 17: 361–367
Hsu GC, Spilburg CA, Bull C, Hoffmann BM (1972) Coboglobins: Heterotropic linkage and the existence of a quaternary structure change upon oxygenation of cobalthemoglobin. Proc Natl Acad Sci USA 69: 2122–2124
Spilburg CA, Hoffmann BM, Petering DH (1972) Coboglobins. Influence of the apoprotein on oxygen binding to cobaltomyoglobin. J Biol Chem 247: 4219–4223
Dickinson LC, Chien JCW (1973) Comparative biological chemistry of cobalt hemoglobin. J Biol Chem 248: 5005–5011
Yonetani T, Yamamoto H, Woodrow III GV (1974) Studies on cobalt myoglobins and hemoglobins. I. Preparation and optical properties of myoglobins and hemoglobins containing cobalt proto-, meso-, and deuteroporphyrins and thermodynamic characterization of their reversible oxygenation. J Biol Chem 249: 682–690
Gersonde K, Twilfer H, Overkamp M (1977) Allostery of monomeric cobalt hemoglobins, 11th FEBS Meeting Copenhagen, Abstracts B2: 458
Gersonde K, Twilfer H, Overkamp M (1978) ESR and oxygen-binding properties of monomeric allosteric cobalt haemoglobins. VIIIth International Conference on Magnetic Resonance in Biological Systems, Nara, Abstracts E10: 106
Chien JCW, Dickinson LC (1972) Electron paramagnetic resonance of single crystal oxycobaltmyoglobin and deoxycobaltmyoglobin. Proc Natl Acad Sci USA 69: 2783–2787
Dickinson LC, Chien JCW (1973) Electron paramagnetic resonance of single crystal deoxycobalthemoglobin. Biochem Biophys Res Commun 51: 587–592
Yonetani T, Yamamoto H, Iizuka T (1974) Studies on cobalt myoglobins and hemoglobins. III. Electron paramagnetic resonance studies of reversible oxygenation of cobalt myoglobins and hemoglobins. J Biol Chem 249: 2168–2174
Gupta RK, Mildvan AS, Yonetani T, Srivastava TS (1975) EPR study of 17O nuclear hyperfine interaction in cobalt-oxyhemoglobin: Conformation of bound oxygen. Biochem Biophys Res Commun 67: 1005–1012
Ikeda-Saito M, Yamamoto H, Yonetani T (1977) Studies on cobalt myoglobins and hemoglobins. Electron paramagnetic resonance of iron-cobalt hybrid hemoglobins and its implication for the heme-heme interaction and for the alkaline Bohr-effect. J Biol Chem 252: 8639–8644
Rossi-Fanelli A, Antonini E, Caputo A (1958) Studies on the structure of hemoglobin. Physicochemical properties of human globin. Biochim Biophys Acta 30: 608–615
Amiconi G, Antonini E, Brunori M, Formanek H, Huber R (1972) Functional properties of native and reconstituted hemoglobins from Chironomus thummi thummi. Eur J Biochem 31: 52–58
Labbe RF, Nishida G (1957) A new method of hemin isolation. Biochim Biophys Acta 26: 437
Morrell DB, Stewart M (1956) The removal of iron from haemins. Austral J Exp Biol 34: 211–218
Taylor JF (1940) Metalloporphyrins: Cobalt and manganese mesoporphyrins in coordination with nitrogeneous bases. J Biol Chem 135: 569–595
Wüthrich K (1970) Structural studies of hemes and hemoproteins by nuclear magnetic resonance spectroscopy. Structure and Bonding, vol 8. Springer, Berlin Heidelberg New York, pp 53–121
Sick H, Gersonde K (1969) Method of continuous registration of O2-binding curves of hemoproteins by means of a diffusion chamber. Anal Biochem 32: 362–376
Sick H, Gersonde K (1972) Theory and application of the diffusion technique for measurement and analysis of O2-binding properties of very autoxidizable hemoproteins. Anal Biochem 47: 46–56
Twilfer H, Gersonde K, Christahl M (1981) Resolution enhancement of EPR spectra using the Fourier transform technique. Analysis of nitrosyl cytochrome c oxidase in frozen solution. J Magn Reson 44: 470–478
Gersonde K, Overkamp M, Sick H, Trittelvitz E, Junge W (1973) Β-Chain allostery in the frozen quaternary T-structure of haemoglobin M Iwate. Eur J Biochem 39: 403–412
Overkamp M (1979) Untersuchungen zur Allosterie in monomeren und dimeren HÄmoglobinen der Zuckmücke Chironomus thummi thummi (Diptera, Insecta). Dissertation, Aachen
La Mar GN, Budd DL, Sick H, Gersonde K (1978) Acid Bohr effects in myoglobin characterized by proton NMR hyperfine shifts and oxygen binding studies. Biochim Biophys Acta 537: 270–283
Imai K, Yonetani T (1975) pH dependence of the Adair constants of human hemoglobin. Nonuniform contribution of successive oxygen bindings to the alkaline Bohr effect. J Biol Chem 250: 2227–2231
Imai K, Yonetani T (1975) Thermodynamical studies of oxygen equilibrium of hemoglobin. Nonuniform heats and entropy changes for the individual oxygenation steps and enthalpy-entropy compensation. J Biol Chem 250: 7093–7098
Tamura M, Woodrow III GV, Yonetani T (1973) Heme-modification studies of myoglobin. II. Ligand binding characteristics of ferric and ferrous myoglobins containing unnatural hemes. Biochim Biophys Acta 317: 34–49
Ikeda-Saito M, Iizuka T, Yamamoto H, Kayne FJ, Yonetani T (1977) Studies on cobalt myoglobins and hemoglobins. Interaction of sperm whale myoglobin and Glycera hemoglobin with molecular oxygen. J Biol Chem 252: 4882–4887
Imai K, Yonetani T, Ikeda-Saito M (1977) Allosteric effects in cobalto haemoglobin as studied by precise oxygen equilibrium measurements. J Mol Biol 109: 83–97
Little GR, Ibers JA (1974) Stereochemistry of cobalt porphyrins. I. The structure and characterization of 2,3,7,8,12,13,17,18-octaaethylporphinato-bis(3-methylpyridine)cobalt(II). J Am Chem Soc 96: 4440–4446
Yamamoto H, Kayne FJ, Yonetani T (1974) Studies on cobalt myoglobins and hemoglobins. II. Kinetic studies of reversible oxygenation of cobalt myoglobins and hemoglobins by the temperature jump relaxation method. J Biol Chem 249: 691–698
Ikeda-Saito M, Brunori M, Yonetani T (1978) Oxygenation and epr spectral properties of Aplysia myoglobins containing cobaltous porphyrins. Biochim Biophys Acta 533: 173–180
Kilmartin JV, Rossi-Bernardi L (1973) Interaction of hemoglobin with hydrogen ions, carbon dioxide, and organic phosphates. Physiol Rev 53: 836–890
Nicola NA, Leach SJ (1977) Structural rearrangements due to ligand binding and haem replacement in myoglobin and leghaemoglobin. Eur J Biochem 78: 133–140
Johnson RN, Bradbury JH, Appleby C (1978) A proton magnetic resonance study of the distal histidine of soybean leghemoglobin. J Biol Chem 253: 2148–2154
Perutz MF (1976) Structure and mechanism of haemoglobin. Br Med Bull 32: 195–208
Lauher JW, Ibers JA (1974) Stereochemistry of cobaltporphyrins. II. The characterization and structure of meso-tetraphenylporphinatobis(imidazole)cobalt(III) acetate monohydrate monochloroformate, (Co(Im)2(TPP)) (O Ac) · H2O · CHCl3. J Am Chem Soc 96: 4447–4452
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gersonde, K., Twilfer, H. & Overkamp, M. Bohr-effect and pH-dependence of electron spin resonance spectra of a cobalt-substituted monomeric insect haemoglobin. Biophys. Struct. Mechanism 8, 189–211 (1982). https://doi.org/10.1007/BF00535459
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00535459