Abstract
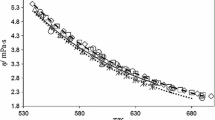
The viscosity of molten sodium chloride (NaCl) has been measured in the temperature range 1083–1248 K by the oscillating-cup method. Although NaCl was considered as one of the standard salts, there existed a systematic deviation among the available data. The present measurement is the redetermination after checking the effects of the meniscus at the liquid surface and of the temperature field.
Similar content being viewed by others
References
G. J. Janz, F. W. Dampier, G. R. Lakshminarayanan, P. K. Lorentz, and R. P. T. Tomkins, Molten Salts: Viscosity Data (NSRDS, NBS, 1968), p. 48.
G. J. Janz, J. Phys. Chem. Ref. Data 9:791 (1980).
G. J. Janz, Proc. 8th Symp. Thermophys. Prop. Vol. II (ASME, New York, 1982), p. 256.
K. Torklep and H. A. Oye, Ber. Bunsenges. Phys. Chem. 83:1 (1979).
Y. Abe, O. Kosugiyama, and A. Nagashima, Ber. Bunsenges. Phys. Chem. 84:1178 (1980).
K. Torklep and H. A. Oye, Ber. Bunsenges. Phys. Chem. 85:814 (1981).
T. Ejima, K. Shimakage, Y. Sata, H. Okuda, N. Kumada, and A. Ishigaki, J. Chem. Soc. Jap. 961 (1982).
T. Ejima, Y. Sato, and E. Takeuchi, 6th Japan. Symp. Thermophys. Prop., Sendai (1985), p. 69.
Y. Abe, O. Kosugiyama, and A. Nagashima, J. Nucl. Mater. 99:173 (1981).
Y. Abe, O. Kosugiyama, H. Mitajima, and A. Nagashima, J. Chem. Soc. Faraday I 76:2531 (1980).
J. Kestin and G. F. Newell, Z. angew. Meth. Phys. 8:433 (1957).
G. S. Kell, J. Chem. Eng. Data 20:97 (1975).
A. Nagashima, J. Phys. Chem. Ref. Data 6:1133 (1977).
M. V. Smirnov, V. A. Khokhlov, and A. A. Antonov, Trudi Inst. Elektrokhim. Ural. Nauch. Tsentr AN SSSR 24:10 (1976).
W. Brockner, K. Grjotheim, T. Ohta, and H. A. Oye, Ber. Bunsenges. Phys. Chem. 79:344 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ito, T., Kojima, N. & Nagashima, A. Redetermination of the viscosity of molten NaCl at elevated temperatures. Int J Thermophys 10, 819–831 (1989). https://doi.org/10.1007/BF00514478
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00514478